Lack of serotonin1B receptor expression leads to age-related motor dysfunction, early onset of brain molecular aging and reduced longevity
- PMID: 17420766
- PMCID: PMC2515886
- DOI: 10.1038/sj.mp.4001990
Lack of serotonin1B receptor expression leads to age-related motor dysfunction, early onset of brain molecular aging and reduced longevity
Abstract
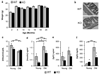
Normal aging of the brain differs from pathological conditions and is associated with increased risk for psychiatric and neurological disorders. In addition to its role in the etiology and treatment of mood disorders, altered serotonin (5-HT) signaling is considered a contributing factor to aging; however, no causative role has been identified in aging. We hypothesized that a deregulation of the 5-HT system would reveal its contribution to age-related processes and investigated behavioral and molecular changes throughout adult life in mice lacking the regulatory presynaptic 5-HT(1B) receptor (5-HT(1B)R), a candidate gene for 5-HT-mediated age-related functions. We show that the lack of 5-HT(1B)R (Htr1b(KO) mice) induced an early age-related motor decline and resulted in decreased longevity. Analysis of life-long transcriptome changes revealed an early and global shift of the gene expression signature of aging in the brain of Htr1b(KO) mice. Moreover, molecular changes reached an apparent maximum effect at 18-months in Htr1b(KO) mice, corresponding to the onset of early death in that group. A comparative analysis with our previous characterization of aging in the human brain revealed a phylogenetic conservation of age-effect from mice to humans, and confirmed the early onset of molecular aging in Htr1b(KO) mice. Potential mechanisms appear independent of known central mechanisms (Bdnf, inflammation), but may include interactions with previously identified age-related systems (IGF-1, sirtuins). In summary, our findings suggest that the onset of age-related events can be influenced by altered 5-HT function, thus identifying 5-HT as a modulator of brain aging, and suggesting age-related consequences to chronic manipulation of 5-HT.
Figures



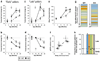

Similar articles
-
Serotonin/dopamine interactions in a hyperactive mouse: reduced serotonin receptor 1B activity reverses effects of dopamine transporter knockout.PLoS One. 2014 Dec 16;9(12):e115009. doi: 10.1371/journal.pone.0115009. eCollection 2014. PLoS One. 2014. PMID: 25514162 Free PMC article.
-
5-HT(1A) receptor sensitivity in 5-HT(1B) receptor KO mice is unaffected by chronic fluvoxamine treatment.Eur J Pharmacol. 2011 Sep 30;667(1-3):250-7. doi: 10.1016/j.ejphar.2011.06.018. Epub 2011 Jun 25. Eur J Pharmacol. 2011. PMID: 21723276
-
A Lack of Serotonin 1B Autoreceptors Results in Decreased Anxiety and Depression-Related Behaviors.Neuropsychopharmacology. 2016 Nov;41(12):2941-2950. doi: 10.1038/npp.2016.109. Epub 2016 Jun 29. Neuropsychopharmacology. 2016. PMID: 27353308 Free PMC article.
-
Experimental gene interaction studies with SERT mutant mice as models for human polygenic and epistatic traits and disorders.Genes Brain Behav. 2003 Dec;2(6):350-64. doi: 10.1046/j.1601-1848.2003.00049.x. Genes Brain Behav. 2003. PMID: 14653307 Review.
-
Behavioral and serotonergic consequences of decreasing or increasing hippocampus brain-derived neurotrophic factor protein levels in mice.Neuropharmacology. 2008 Nov;55(6):1006-14. doi: 10.1016/j.neuropharm.2008.08.001. Epub 2008 Aug 12. Neuropharmacology. 2008. PMID: 18761360 Review.
Cited by
-
Synaptic underpinnings of altered hippocampal function in glutaminase-deficient mice during maturation.Hippocampus. 2012 May;22(5):1027-39. doi: 10.1002/hipo.22014. Epub 2012 Mar 19. Hippocampus. 2012. PMID: 22431402 Free PMC article.
-
Hospitalisation for COVID-19 predicts long lasting cerebrovascular impairment: A prospective observational cohort study.Neuroimage Clin. 2022;36:103253. doi: 10.1016/j.nicl.2022.103253. Epub 2022 Nov 7. Neuroimage Clin. 2022. PMID: 36451358 Free PMC article.
-
Age-dependent increase in Kalirin-9 and Kalirin-12 transcripts in human orbitofrontal cortex.Eur J Neurosci. 2016 Oct;44(7):2483-2492. doi: 10.1111/ejn.13351. Epub 2016 Aug 18. Eur J Neurosci. 2016. PMID: 27471199 Free PMC article.
-
Transcriptomic approach predicts a major role for transforming growth factor beta type 1 pathway in L-Dopa-induced dyskinesia in parkinsonian rats.Genes Brain Behav. 2020 Nov;19(8):e12690. doi: 10.1111/gbb.12690. Epub 2020 Sep 11. Genes Brain Behav. 2020. PMID: 32741046 Free PMC article.
-
HTR1B as a risk profile maker in psychiatric disorders: a review through motivation and memory.Eur J Clin Pharmacol. 2010 Jan;66(1):5-27. doi: 10.1007/s00228-009-0724-6. Epub 2009 Oct 7. Eur J Clin Pharmacol. 2010. PMID: 19809810 Review.
References
-
- Sowell ER, Peterson BS, Thompson PM, Welcome SE, Henkenius AL, Toga AW. Mapping cortical change across the human life span. Nat Neurosci. 2003;6:309–315. - PubMed
-
- Terry RD, DeTeresa R, Hansen LA. Neocortical cell counts in normal human adult aging. Ann Neurol. 1987;21:530–539. - PubMed
-
- Bertoni-Freddari C, Fattoretti P, Paoloni R, Caselli U, Galeazzi L, Meier-Ruge W. Synaptic structural dynamics and aging. Gerontology. 1996;42:170–180. - PubMed
-
- Pakkenberg B, Gundersen HJ. Neocortical neuron number in humans: effect of sex and age. J Comp Neurol. 1997;384:312–320. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

