Calcium and arrhythmogenesis
- PMID: 17429038
- PMCID: PMC4332537
- DOI: 10.1152/physrev.00011.2006
Calcium and arrhythmogenesis
Abstract
Triggered activity in cardiac muscle and intracellular Ca2+ have been linked in the past. However, today not only are there a number of cellular proteins that show clear Ca2+ dependence but also there are a number of arrhythmias whose mechanism appears to be linked to Ca2+-dependent processes. Thus we present a systematic review of the mechanisms of Ca2+ transport (forward excitation-contraction coupling) in the ventricular cell as well as what is known for other cardiac cell types. Second, we review the molecular nature of the proteins that are involved in this process as well as the functional consequences of both normal and abnormal Ca2+ cycling (e.g., Ca2+ waves). Finally, we review what we understand to be the role of Ca2+ cycling in various forms of arrhythmias, that is, those associated with inherited mutations and those that are acquired and resulting from reentrant excitation and/or abnormal impulse generation (e.g., triggered activity). Further solving the nature of these intricate and dynamic interactions promises to be an important area of research for a better recognition and understanding of the nature of Ca2+ and arrhythmias. Our solutions will provide a more complete understanding of the molecular basis for the targeted control of cellular calcium in the treatment and prevention of such.
Figures


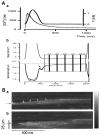




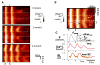
References
-
- Aggarwal R, Boyden PA. Diminished calcium and barium currents in myocytes surviving in the epicardial border zone of the 5 day infarcted canine heart. Circ Res. 1995;77:1180–1191. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

