Interactions between estrogen- and Ah-receptor signalling pathways in primary culture of salmon hepatocytes exposed to nonylphenol and 3,3',4,4'-tetrachlorobiphenyl (congener 77)
- PMID: 17433103
- PMCID: PMC1855068
- DOI: 10.1186/1476-5926-6-2
Interactions between estrogen- and Ah-receptor signalling pathways in primary culture of salmon hepatocytes exposed to nonylphenol and 3,3',4,4'-tetrachlorobiphenyl (congener 77)
Abstract
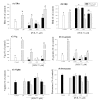
Background: The estrogenic and xenobiotic biotransformation gene expressions are receptor-mediated processes that are ligand structure-dependent interactions with estrogen-receptor (ER) and aryl hydrocarbon receptor (AhR), probably involving all subtypes and other co-factors. The anti-estrogenic activities of AhR agonists have been reported. In teleost fish, exposure to AhR agonists has been associated with reduced Vtg synthesis or impaired gonadal development in both in vivo- and in vitro studies. Inhibitory AhR and ER cross-talk have also been demonstrated in breast cancer cells, rodent uterus and mammary tumors. Previous studies have shown that AhR-agonists potentiate xenoestrogen-induced responses in fish in vivo system. Recently, several studies have shown that AhR-agonists directly activate ER alpha and induce estrogenic responses in mammalian in vitro systems. In this study, two separate experiments were performed to study the molecular interactions between ER and AhR signalling pathways using different concentration of PCB-77 (an AhR-agonist) and time factor, respectively. Firstly, primary Atlantic salmon hepatocytes were exposed to nonylphenol (NP: 5 microM--an ER agonist) singly or in combination with 0.001, 0.01 and 1 microM PCB-77 and sampled at 48 h post-exposure. Secondly, hepatocytes were exposed to NP (5 microM) or PCB-77 (1 microM) singly or in combination for 12, 24, 48 and 72 h. Samples were analyzed using a validated real-time PCR for genes in the ER pathway or known to be NP-responsive and AhR pathway or known to be PCB-77 responsive.
Results: Our data showed a reciprocal inhibitory interaction between NP and PCB-77. PCB-77 produced anti-NP-mediated effect by decreasing the mRNA expression of ER-responsive genes. NP produced anti-AhR mediated effect or as inhibitor of AhR alpha, AhRR, ARNT, CYP1A1 and UDPGT expression. A novel aspect of the present study is that low (0.001 microM) and medium (0.01 microM) PCB-77 concentrations increased ER alpha mRNA expression above control and NP exposed levels, and at 12 h post-exposure, PCB-77 exposure alone produced significant elevation of ER alpha, ER beta and Zr-protein expressions above control levels.
Conclusion: The findings in the present study demonstrate a complex mode of ER-AhR interactions that were dependent on time of exposure and concentration of individual chemicals (NP and PCB-77). This complex mode of interaction is further supported by the effect of PCB-77 on ER alpha and ER beta (shown as increase in transcription) with no concurrent activation of Vtg (but Zr-protein) response. These complex interactions between two different classes of ligand-activated receptors provide novel mechanistic insights on signalling pathways. Therefore, the degree of simultaneous interactions between the ER and AhR gene transcripts demonstrated in this study supports the concept of cross-talk between these signalling pathways.
Figures



References
-
- Gonzalez FJ, Fernandez-Salguero P. The aryl hydrocarbon receptor: studies using the AHR-null mice. Drug Metab Dispos. 1998;26:1194–1198. - PubMed
-
- Mommsen PT, Walsh PJ. Vitellogenesis and oocyte assembly. In: Hoar WS, Randall DJ, Donaldson EM, editor. Fish Physiology. XIA. New York: Academic Press; 1988. pp. 347–406.
-
- Oppen-Berntsen DO, Gram-Jensen E, Walther BT. Zona radiata proteins are synthesized by rainbow trout (Oncorhynchus mykiss) hepatocytes in response to oestradiol-17 beta. J Endocrinol. 1992;135:293–302. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
