Short telomeres limit tumor progression in vivo by inducing senescence
- PMID: 17433785
- PMCID: PMC1945093
- DOI: 10.1016/j.ccr.2007.02.026
Short telomeres limit tumor progression in vivo by inducing senescence
Abstract
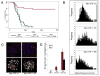
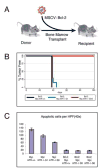
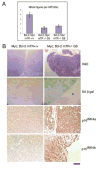
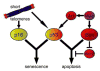
Telomere maintenance is critical for cancer progression. To examine mechanisms of tumor suppression induced by short telomeres, we crossed mice deficient for the RNA component of telomerase, mTR(-/-), with Emu-myc transgenic mice, an established model of Burkitt's lymphoma. Short telomeres suppressed tumor formation in Emu-myc transgenic animals. Expression of Bcl2 blocked apoptosis in tumor cells, but surprisingly, mice with short telomeres were still resistant to tumor formation. Staining for markers of cellular senescence showed that pretumor cells induced senescence in response to short telomeres. Loss of p53 abrogated the short telomere response. This study provides in vivo evidence for the existence of a p53-mediated senescence mechanism in response to short telomeres that suppresses tumorigenesis.
Figures


Comment in
-
Telomeres limit cancer growth by inducing senescence: long-sought in vivo evidence obtained.Cancer Cell. 2007 May;11(5):389-91. doi: 10.1016/j.ccr.2007.04.014. Cancer Cell. 2007. PMID: 17482128
References
-
- Adams JM, Harris AW, Pinkert CA, Corcoran LM, Alexander WS, Cory S, Palmiter RD, Brinster RL. The c-myc oncogene driven by immunoglobulin enhancers induces lymphoid malignancy in transgenic mice. Nature. 1985;318:533–538. - PubMed
-
- Artandi SE, Chang S, Lee SL, Alson S, Gottlieb GJ, Chin L, DePinho RA. Telomere dysfunction promotes non-reciprocal translocations and epithelial cancers in mice. Nature. 2000;406:641–645. - PubMed
-
- Blasco MA, Lee HW, Hande MP, Samper E, Lansdorp PM, DePinho RA, Greider CW. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell. 1997;91:25–34. - PubMed
-
- Bodnar AG, Ouellette M, Frolkis M, Holt SE, Chiu CP, Morin GB, Harley CB, Shay JW, Lichtsteiner S, Wright WE. Extension of life-span by introduction of telomerase into normal human cells. Science. 1998;279:349–352. - PubMed
-
- Braig M, Lee S, Loddenkemper C, Rudolph C, Peters AH, Schlegelberger B, Stein H, Dorken B, Jenuwein T, Schmitt CA. Oncogene-induced senescence as an initial barrier in lymphoma development. Nature. 2005;436:660–665. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

