Dihydroxyoctadecamonoenoate esters inhibit the neutrophil respiratory burst
- PMID: 17435320
- PMCID: PMC1904342
- DOI: 10.1007/s12038-007-0028-x
Dihydroxyoctadecamonoenoate esters inhibit the neutrophil respiratory burst
Abstract
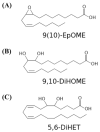
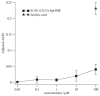
The leukotoxins [9(10)-and 12(13)-EpOME] are produced by activated inflammatory leukocytes such as neutrophils. High EpOME levels are observed in disorders such as acute respiratory distress syndrome and in patients with extensive burns.Although the physiological significance of the EpOMEs remains poorly understood,in some systems, the EpOMEs act as a protoxin,with their corresponding epoxide hydrolase metabolites,9,10-and 12,13-DiHOME, specifically exerting toxicity.Both the EpOMEs and the DiHOMEs were also recently shown to have neutrophil chemotactic activity.We evaluated whether the neutrophil respiratory burst,a surge of oxidant production thought to play an important role in limiting certain bacterial and fungal infections,is modulated by members of the EpOME metabolic pathway.We present evidence that the DiHOMEs suppress the neutrophil respiratory burst by a mechanism distinct from that of respiratory burst inhibitors such as cyclosporin H or lipoxin A4,which inhibit multiple aspects of neutrophil activation.
Figures





Similar articles
-
Cytochrome P450-derived linoleic acid metabolites EpOMEs and DiHOMEs: a review of recent studies.J Nutr Biochem. 2020 Dec;86:108484. doi: 10.1016/j.jnutbio.2020.108484. Epub 2020 Aug 20. J Nutr Biochem. 2020. PMID: 32827665 Free PMC article. Review.
-
Insect immune resolution with EpOME/DiHOME and its dysregulation by their analogs leading to pathogen hypersensitivity.bioRxiv [Preprint]. 2023 Jul 7:2023.07.07.548078. doi: 10.1101/2023.07.07.548078. bioRxiv. 2023. Update in: Insect Biochem Mol Biol. 2024 May;168:104104. doi: 10.1016/j.ibmb.2024.104104. PMID: 37461499 Free PMC article. Updated. Preprint.
-
Formyl-methionyl-leucyl-phenylalanine and a calcium ionophore A23187 reverse the inhibition of phorbol myristate acetate-induced oxidative burst by linoleic and oleic acid anilides.Toxicology. 1996 Jun 17;110(1-3):39-45. doi: 10.1016/0300-483x(96)03320-3. Toxicology. 1996. PMID: 8658558
-
EpOMEs act as immune suppressors in a lepidopteran insect, Spodoptera exigua.Sci Rep. 2020 Nov 19;10(1):20183. doi: 10.1038/s41598-020-77325-2. Sci Rep. 2020. PMID: 33214688 Free PMC article.
-
[Modulation of the oxidative burst of human neutrophils by pro- and anti-inflammatory cytokines].Pathol Biol (Paris). 1996 Jan;44(1):36-41. Pathol Biol (Paris). 1996. PMID: 8734298 Review. French.
Cited by
-
Untargeted Metabolomic Analysis Reveals Plasma Differences between Mares with Endometritis and Healthy Ones.Animals (Basel). 2024 Jun 29;14(13):1933. doi: 10.3390/ani14131933. Animals (Basel). 2024. PMID: 38998045 Free PMC article.
-
Role of endogenous TRPV1 agonists in a postburn pain model of partial-thickness injury.Pain. 2013 Nov;154(11):2512-2520. doi: 10.1016/j.pain.2013.07.040. Epub 2013 Jul 26. Pain. 2013. PMID: 23891895 Free PMC article.
-
Oxidized Lipids in Persistent Pain States.Front Pharmacol. 2019 Oct 15;10:1147. doi: 10.3389/fphar.2019.01147. eCollection 2019. Front Pharmacol. 2019. PMID: 31680947 Free PMC article. Review.
-
Maximal exercise and plasma cytochrome P450 and lipoxygenase mediators: a lipidomics study.Physiol Rep. 2019 Jul;7(13):e14165. doi: 10.14814/phy2.14165. Physiol Rep. 2019. PMID: 31304687 Free PMC article.
-
Metabolic profiling of plasma from cardiac surgical patients concurrently administered with tranexamic acid: DI-SPME-LC-MS analysis.J Pharm Anal. 2014 Feb;4(1):6-13. doi: 10.1016/j.jpha.2013.03.002. Epub 2013 Apr 11. J Pharm Anal. 2014. PMID: 29403864 Free PMC article.
References
-
- Babior BM. NADPH oxidase: an update. Blood. 1999;93:1464–1476. - PubMed
-
- Badwey JA, Curnutte JT, Karnovsky ML. cis-Polyunsaturated fatty acids induce high levels of superoxide production by human neutrophils. J Biol Chem. 1981;256:12640–12643. - PubMed
-
- Badwey JA, Curnutte JT, Robinson JM, Berde CB, Karnovsky MJ, Karnovsky ML. Effects of free fatty acids on release of superoxide and on change of shape by human neutrophils reversibility by albumin. J Biol Chem. 1984;259:7870–7877. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

