Expression and cellular localization of molecules of the gp82 family in Trypanosoma cruzi metacyclic trypomastigotes
- PMID: 17438027
- PMCID: PMC1932952
- DOI: 10.1128/IAI.00262-07
Expression and cellular localization of molecules of the gp82 family in Trypanosoma cruzi metacyclic trypomastigotes
Abstract
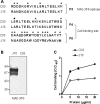
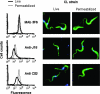
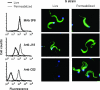
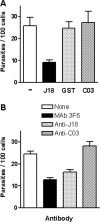
A member of the Trypanosoma cruzi gp82 family, expressed on metacyclic trypomastigote surface and identified by monoclonal antibody (MAb) 3F6, plays a key role in host cell invasion. Apart from the gp82 defined by MAb 3F6, no information is available on members of this protein family. From cDNA clones encoding gp82 proteins sharing 59.1% sequence identity, we produced the recombinant proteins J18 and C03, the former containing and the latter lacking the epitope for MAb 3F6. Polyclonal antibodies to J18 and C03 proteins were generated and used, along with MAb 3F6, to analyze the expression and cellular localization of gp82 family members in metacyclic forms of CL and G strains, which belong to highly divergent genetic groups. By two-dimensional gel electrophoresis and immunoblotting, molecules of 82 to 86 kDa, focusing at pH 4.6 to 5.4, and molecules of 72 to 88 kDa, focusing at pH 4.9 to 5.7, were visualized in CL and G strains, respectively. Flow cytometry and microscopic analysis revealed in both strains similar expression of MAb 3F6-reactive gp82 in live and permeabilized parasites, indicating its surface localization. The reaction of live parasites of both strains with anti-J18 antibodies was weaker than with MAb 3F6 and was increased by permeabilization. Anti-C03 antibodies bound predominantly to flagellar components in permeabilized G strain parasites, but in the CL strain the flagellum was not the preferential target for these antibodies. Host cell invasion of metacyclic forms was inhibited by J18 protein, as well as by MAb 3F6 and anti-J18 antibodies, but not by C03 protein or anti-C03 antibodies.
Figures


References
-
- Araya, J. E., M. I. Cano, N. Yoshida, N., and J. Franco da Silveira. 1994. Cloning and characterization of a gene for the stage-specific 82-kDa surface antigen of metacyclic trypomastigotes of Trypanosoma cruzi. Mol. Biochem. Parasitol. 65:161-169. - PubMed
-
- Atwood, J. A., D. B. Weatherly, T. A Minning, B. Bundy, C. Cavola, F. R. Operdoes, R. Orlando, and R. L. Tarleton. 2005. The Trypanosoma cruzi proteome. Science 309:473-476. - PubMed
-
- Bendtsen, J. D., H. Nielsen, G. von Heijne, and S. Brunak. 2004. Improved prediction of signal peptides: SignalP 3.0. J. Mol. Biol. 340:783-795. - PubMed
-
- Brener, Z., and E. Chiari. 1963. Variações morfológicas observadas em diferentes amostras de Trypanosoma cruzi. Rev. Inst. Méd. Trop. São Paulo 5:220-224. - PubMed
-
- Briones, M. R. S., R. P. Souto, B. S. Stolf, and B. Zingales. 1999. The evolution of two Trypanosoma cruzi subgroups inferred from rRNA genes can be correlated with the interchange of American mammalian faunas in the Cenozoic and has implications to pathogenicity and host specificity. Mol. Biochem. Parasitol. 104:219-232. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources

