Activation of TRPA1 channel facilitates excitatory synaptic transmission in substantia gelatinosa neurons of the adult rat spinal cord
- PMID: 17442829
- PMCID: PMC6672326
- DOI: 10.1523/JNEUROSCI.0557-07.2007
Activation of TRPA1 channel facilitates excitatory synaptic transmission in substantia gelatinosa neurons of the adult rat spinal cord
Abstract
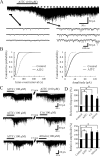
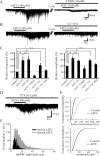
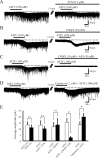
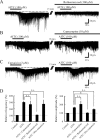
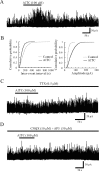
TRPA1 is expressed in primary sensory neurons and hair cells, and it is proposed to be activated by cold stimuli, mechanical stimuli, or pungent ingredients. However, its role in regulating synaptic transmission has never been documented yet. In the present study, we examined whether activation of the TRPA1 channels affects synaptic transmission in substantia gelatinosa (SG) neurons of adult rat spinal cord slices by using the whole-cell patch-clamp technique. A chief ingredient of mustard oil, allyl isothiocyanate (AITC), superfused for 2 min markedly increased the frequency and amplitude of spontaneous EPSCs (sEPSCs), which was accompanied by an inward current. Similar actions were produced by cinnamaldehyde and allicin. The AITC-induced increases in sEPSC frequency and amplitude were resistant to tetrodotoxin (TTX) and La3+, whereas being significantly reduced in extent in a Ca2+-free bath solution. In the presence of glutamate receptor antagonists CNQX and AP5, AITC did not generate any synaptic activities. The AITC-induced increases in sEPSC frequency and amplitude were reduced by ruthenium red, whereas being unaffected by capsazepine. AITC also increased the frequency and amplitude of spontaneous inhibitory postsynaptic currents; this AITC action was abolished in the presence of TTX or glutamate receptor antagonists. These results indicate that TRPA1 appears to be localized not only at presynaptic terminals on SG neurons to enhance glutamate release, but also in terminals of primary afferents innervating onto spinal inhibitory interneurons, which make synapses with SG neurons. This central modulation of sensory signals may be associated with physiological and pathological pain sensations.
Figures
References
-
- Bandell M, Story GM, Hwang SW, Viswanath V, Eid SR, Petrus MJ, Earley TJ, Patapoutian A. Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron. 2004;41:849–857. - PubMed
-
- Bautista DM, Jordt S-E, Nikai T, Tsuruda PR, Read AJ, Poblete J, Yamoah EN, Basbaum AI, Julius D. TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell. 2006;124:1269–1282. - PubMed
-
- Clapham DE. TRP channels as cellular sensors. Nature. 2003;426:517–524. - PubMed
-
- Corey DP, García-Añoveros J, Holt JR, Kwan KY, Lin S-Y, Vollrath MA, Amalfitano A, Cheung EL-M, Derfler BH, Duggan A, Géléoc GSG, Gray PA, Hoffman MP, Rehm HL, Tamasauskas D, Zhang D-S. TRPA1 is a candidate for the mechanosensitive transduction channel of vertebrate hair cells. Nature. 2004;432:723–730. - PubMed
-
- Gu JG, MacDermott AB. Activation of ATP P2X receptors elicits glutamate release from sensory neuron synapses. Nature. 1997;389:749–753. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
