Radial spoke protein 44 (human meichroacidin) is an axonemal alloantigen of sperm and cilia
- PMID: 17451891
- PMCID: PMC1935023
- DOI: 10.1016/j.gene.2007.02.031
Radial spoke protein 44 (human meichroacidin) is an axonemal alloantigen of sperm and cilia
Abstract
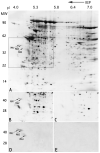
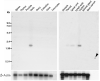
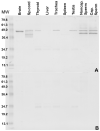
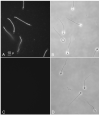
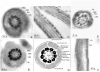
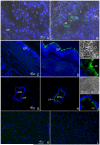
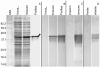
To identify novel sperm alloantigens relevant to immune infertility, sera from infertile men containing antisperm antibodies (ASA) were employed on 2-D immunoblots of human sperm proteins. An immunoreactive protein spot (MW: 44 kDa, pI: 4.5) was microsequenced and the related cDNA was cloned yielding a 309 amino acid sequence corresponding to a gene currently annotated in Genbank as TSGA2 homolog (mouse) to signify 'testis specific gene A2'. In Genbank the protein deduced from this gene is currently named human meichroacidin, an orthologue of meichroacidin previously identified in mouse spermatocytes. Human TSGA2 mapped to chromosome 21q22.3. Human meichroacidin (hMCA) contained a single potential tyrosine phosphorylation site and five casein kinase phosporylation motifs. The N-terminus contained a Membrane Occupation Recognition Nexus (MORN) motif found in the lipid kinase-phosphatidylinositol 4-phosphate 5-kinase (PIP5K) family and junctophilins. However hMCA lacked the characteristic kinase homology domain of PIP5K. Northern blot analysis revealed 1.5 kb hMCA transcripts in testis and trachea with lower levels in thyroid and spinal cord. A semi-quantitative reverse transcription polymerase chain reaction (RT-PCR) analysis demonstrated occurrence of the mRNA messages in all the ciliated tissues tested with highest levels of messages in testis and trachea. Western blot analysis showed the presence of hMCA protein in brain, thyroid and trachea at the identical mass, 44 kDa, as in human testis. However, this immunoreactive pattern differed from that of sperm in which a 38 kDa form was also evident suggesting that hMCA undergoes proteolytic processing. In human testis, hMCA localized to the tails of developing spermatids and did not localize to the nucleus of either spermatocytes or spermatids. EM immunocytochemistry localized hMCA within the radial spokes of the axonemal complex of the sperm flagellum, and immunofluorescence studies revealed h-meichroacidin in the cilia of epithelial cells in the trachea and ependyma. Bioinformatic identification of orthologues of meichroacidin in several lower organisms including ciliates and flagellates suggest the protein plays a role in flagellar motility across phyla. We propose the term radial spoke protein 44 as an accurate designation, preferable to human meichroacidin because it denotes the restricted localization of the protein to the radial spokes of the axonemes of both sperm and cilia. Further, since the human gene is expressed in brain, thyroid, trachea and lung in addition to testis, we suggest that the gene name be changed from TSGA2 [testis specific gene A2] to radial spoke protein 44 [RSP44].
Figures



References
-
- Anderson RA, Boronenkov IV, Doughman SD, Kunz J, Loijens JC. Phosphatidylinositol phosphate kinases, a multifaceted family of signaling enzymes. J Biol Chem. 1999;274:9907–9910. - PubMed
-
- Bouchard P, Cosson MP. Molecular basis of sperm movement. Ann. Endocrinol. (Paris) 1981;42:398–406. - PubMed
-
- Bronson RA, Cooper GW, Rosenfeld DL. Correlation between regional specificity of antisperm antibodies to the spermatozoan surface and complement-mediated sperm immobilization. Am. J. Reprod. Immunol. 1982;2:222–224. - PubMed
-
- Bronson RA, Tung KSK. Human sperm antibodies and their detection. In: Rose NR, de Macario ER, editors. Am. Soc. for Microbiol. 4th edition Manual of Clinical Laboratory Immunology; Washington, DC: 1992. pp. 1013–1017.
-
- Caron CP, Saling PM. Sperm antigens and immunological interference of fertilization. In: Wassarman PM, editor. Elements of Mammalian Fertilization. CRC Press; Boca Raton, FL: 1991. pp. 147–176.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

