Toll-like receptor 2-dependent NF-kappaB activation is involved in nontypeable Haemophilus influenzae-induced monocyte chemotactic protein 1 up-regulation in the spiral ligament fibrocytes of the inner ear
- PMID: 17452470
- PMCID: PMC1932924
- DOI: 10.1128/IAI.01886-06
Toll-like receptor 2-dependent NF-kappaB activation is involved in nontypeable Haemophilus influenzae-induced monocyte chemotactic protein 1 up-regulation in the spiral ligament fibrocytes of the inner ear
Abstract
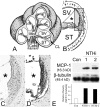
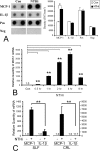
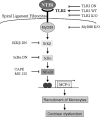
Inner ear dysfunction secondary to chronic otitis media (OM), including high-frequency sensorineural hearing loss or vertigo, is not uncommon. Although chronic middle ear inflammation is believed to cause inner ear dysfunction by entry of OM pathogen components or cytokines from the middle ear into the inner ear, the underlying mechanisms are not well understood. Previously, we demonstrated that the spiral ligament fibrocyte (SLF) cell line up-regulates monocyte chemotactic protein 1 (MCP-1) expression after treatment with nontypeable Haemophilus influenzae (NTHI), one of the most common OM pathogens. We hypothesized that the SLF-derived MCP-1 plays a role in inner ear inflammation secondary to OM that is responsible for hearing loss and dizziness. The purpose of this study was to investigate the signaling pathway involved in NTHI-induced MCP-1 up-regulation in SLFs. Here we show for the first time that NTHI induces MCP-1 up-regulation in the SLFs via Toll-like receptor 2 (TLR2)-dependent activation of NF-kappaB. TLR2(-/-)- and MyD88(-/-)-derived SLFs revealed involvement of TLR2 and MyD88 in NTHI-induced MCP-1 up-regulation. Studies using chemical inhibitors and dominant-negative constructs demonstrated that it is mediated by the IkappaKbeta-dependent IkappaBalpha phosphorylation and NTHI-induced NF-kappaB nuclear translocation. Furthermore, we demonstrated that the binding of NF-kappaB to the enhancer region of MCP-1 is involved in this up-regulation. In addition, we have identified a potential NF-kappaB motif that is responsive and specific to certain NTHI molecules or ligands. Further studies are necessary to reveal specific ligands of NTHI that activate host receptors. These results may provide us with new therapeutic strategies for prevention of inner ear dysfunction secondary to chronic middle ear inflammation.
Figures



Similar articles
-
Spiral ligament fibrocyte-derived MCP-1/CCL2 contributes to inner ear inflammation secondary to nontypeable H. influenzae-induced otitis media.BMC Infect Dis. 2010 Oct 28;10:314. doi: 10.1186/1471-2334-10-314. BMC Infect Dis. 2010. PMID: 21029462 Free PMC article.
-
ERK2-dependent activation of c-Jun is required for nontypeable Haemophilus influenzae-induced CXCL2 upregulation in inner ear fibrocytes.J Immunol. 2012 Apr 1;188(7):3496-505. doi: 10.4049/jimmunol.1103182. Epub 2012 Feb 29. J Immunol. 2012. PMID: 22379036 Free PMC article.
-
Spiral ligament fibrocytes release chemokines in response to otitis media pathogens.Acta Otolaryngol. 2006 Jun;126(6):564-9. doi: 10.1080/00016480500452525. Acta Otolaryngol. 2006. PMID: 16720438
-
[Regulation of expression, function, and inflammatory responses of innate immune receptor Toll-like receptor-2 (TLR2) during inflammatory responses against infection].Yakugaku Zasshi. 2013;133(12):1401-9. doi: 10.1248/yakushi.13-00208. Yakugaku Zasshi. 2013. PMID: 24292189 Review. Japanese.
-
The adhesins of non-typeable Haemophilus influenzae.Expert Rev Anti Infect Ther. 2018 Mar;16(3):187-196. doi: 10.1080/14787210.2018.1438263. Epub 2018 Feb 21. Expert Rev Anti Infect Ther. 2018. PMID: 29415569 Review.
Cited by
-
Correlative mRNA and protein expression of middle and inner ear inflammatory cytokines during mouse acute otitis media.Hear Res. 2015 Aug;326:49-58. doi: 10.1016/j.heares.2015.04.006. Epub 2015 Apr 25. Hear Res. 2015. PMID: 25922207 Free PMC article.
-
IL-10/HMOX1 signaling modulates cochlear inflammation via negative regulation of MCP-1/CCL2 expression in cochlear fibrocytes.J Immunol. 2015 Apr 15;194(8):3953-61. doi: 10.4049/jimmunol.1402751. Epub 2015 Mar 16. J Immunol. 2015. PMID: 25780042 Free PMC article.
-
Spiral ligament fibrocyte-derived MCP-1/CCL2 contributes to inner ear inflammation secondary to nontypeable H. influenzae-induced otitis media.BMC Infect Dis. 2010 Oct 28;10:314. doi: 10.1186/1471-2334-10-314. BMC Infect Dis. 2010. PMID: 21029462 Free PMC article.
-
A High-Fat Diet Induces Low-Grade Cochlear Inflammation in CD-1 Mice.Int J Mol Sci. 2022 May 6;23(9):5179. doi: 10.3390/ijms23095179. Int J Mol Sci. 2022. PMID: 35563572 Free PMC article.
-
Involvement of Ubiquitin-Editing Protein A20 in Modulating Inflammation in Rat Cochlea Associated with Silver Nanoparticle-Induced CD68 Upregulation and TLR4 Activation.Nanoscale Res Lett. 2016 Dec;11(1):240. doi: 10.1186/s11671-016-1430-9. Epub 2016 May 4. Nanoscale Res Lett. 2016. PMID: 27142878 Free PMC article.
References
-
- Adachi, O., T. Kawai, K. Takeda, M. Matsumoto, H. Tsutsui, M. Sakagami, K. Nakanishi, and S. Akira. 1998. Targeted disruption of the MyD88 gene results in loss of IL-1- and IL-18-mediated function. Immunity 9:143-150. - PubMed
-
- Aderem, A., and R. J. Ulevitch. 2000. Toll-like receptors in the induction of the innate immune response. Nature 406:782-787. - PubMed
-
- Agrawal, S., M. Husein, and D. MacRae. 2005. Complications of otitis media: an evolving state. J. Otolaryngol 34(Suppl. 1):S33-S39. - PubMed
-
- Akira, S., and K. Takeda. 2004. Toll-like receptor signalling. Nat. Rev. Immunol. 4:499-511. - PubMed
-
- Akira, S., M. Yamamoto, and K. Takeda. 2003. Role of adapters in Toll-like receptor signalling. Biochem. Soc. Trans. 31:637-642. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

