Adapter protein SH2-Bbeta stimulates actin-based motility of Listeria monocytogenes in a vasodilator-stimulated phosphoprotein (VASP)-dependent fashion
- PMID: 17452473
- PMCID: PMC1932951
- DOI: 10.1128/IAI.00214-07
Adapter protein SH2-Bbeta stimulates actin-based motility of Listeria monocytogenes in a vasodilator-stimulated phosphoprotein (VASP)-dependent fashion
Abstract
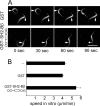
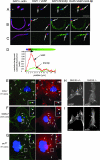
SH2-Bbeta (Src homology 2 Bbeta) is an adapter protein that is required for maximal growth hormone-dependent actin reorganization in membrane ruffling and cell motility. Here we show that SH2-Bbeta is also required for maximal actin-based motility of Listeria monocytogenes. SH2-Bbeta localizes to Listeria-induced actin tails and increases the rate of bacterial propulsion in infected cells and in cell extracts. Furthermore, Listeria motility is decreased in mouse embryo fibroblasts from SH2-B(-/-) mice. Both recruitment of SH2-Bbeta to Listeria and SH2-Bbeta stimulation of actin-based propulsion require the vasodilator-stimulated phosphoprotein (VASP), which binds ActA at the surfaces of Listeria cells and enhances bacterial actin-based motility. SH2-Bbeta enhances actin-based movement of ActA-coated beads in a biomimetic actin-based motility assay, provided that VASP is present. In vitro binding assays show that SH2-Bbeta binds ActA but not VASP; however, binding to ActA is greater in the presence of VASP. Because VASP also plays an essential regulatory role in actin-based processes in eukaryotic cells, the present results provide mechanistic insight into the functions of both SH2-Bbeta and VASP in motility and also increase our understanding of the fundamental mechanism by which Listeria spreads.
Figures





References
-
- Bear, J. E., J. J. Loureiro, I. Libova, R. Fassler, J. Wehland, and F. B. Gertler. 2000. Negative regulation of fibroblast motility by Ena/VASP proteins. Cell 101:717-728. - PubMed
-
- Bear, J. E., T. M. Svitkina, M. Krause, D. A. Schafer, J. J. Loureiro, G. A. Strasser, I. V. Maly, O. Y. Chaga, J. A. Cooper, G. G. Borisy, and F. B. Gertler. 2002. Antagonism between Ena/VASP proteins and actin filament capping regulates fibroblast motility. Cell 109:509-521. - PubMed
-
- Boujemaa-Paterski, R., E. Gouin, G. Hansen, S. Samarin, C. Le Clainche, D. Didry, P. Dehoux, P. Cossart, C. Kocks, M. F. Carlier, and D. Pantaloni. 2001. Listeria protein ActA mimics WASp family proteins: it activates filament barbed end branching by Arp2/3 complex. Biochemistry 40:11390-11404. - PubMed
-
- Cicchetti, G., P. Maurer, P. Wagener, and C. Kocks. 1999. Actin and phosphoinositide binding by the ActA protein of the bacterial pathogen Listeria monocytogenes. J. Biol. Chem. 274:33616-33626. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

