Fatal gastrointestinal obstruction and hypertension in mice lacking nitric oxide-sensitive guanylyl cyclase
- PMID: 17452643
- PMCID: PMC1863512
- DOI: 10.1073/pnas.0609778104
Fatal gastrointestinal obstruction and hypertension in mice lacking nitric oxide-sensitive guanylyl cyclase
Abstract
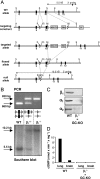
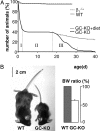
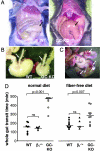
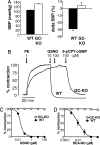
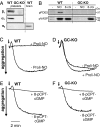
The signaling molecule nitric oxide (NO), first described as endothelium-derived relaxing factor (EDRF), acts as physiological activator of NO-sensitive guanylyl cyclase (NO-GC) in the cardiovascular, gastrointestinal, and nervous systems. Besides NO-GC, other NO targets have been proposed; however, their particular contribution still remains unclear. Here, we generated mice deficient for the beta1 subunit of NO-GC, which resulted in complete loss of the enzyme. GC-KO mice have a life span of 3-4 weeks but then die because of intestinal dysmotility; however, they can be rescued by feeding them a fiber-free diet. Apparently, NO-GC is absolutely vital for the maintenance of normal peristalsis of the gut. GC-KO mice show a pronounced increase in blood pressure, underlining the importance of NO in the regulation of smooth muscle tone in vivo. The lack of an NO effect on aortic relaxation and platelet aggregation confirms NO-GC as the only NO target regulating these two functions, excluding cGMP-independent mechanisms. Our knockout model completely disrupts the NO/cGMP signaling cascade and provides evidence for the unique role of NO-GC as NO receptor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

