Nkx2.2 regulates beta-cell function in the mature islet
- PMID: 17456846
- PMCID: PMC2805073
- DOI: 10.2337/db06-1766
Nkx2.2 regulates beta-cell function in the mature islet
Abstract
Nkx2.2 is a homeodomain transcription factor that is critical for pancreatic endocrine cell specification and differentiation in the developing mouse embryo. The purpose of this study was to determine whether Nkx2.2 is also required for the maintenance and function of the mature beta-cell in the postnatal islet. We have demonstrated previously that a repressor derivative of Nkx2.2 can functionally substitute for endogenous Nkx2.2 to fully restore alpha- and immature beta-cells in the embryonic islet; however, Nkx2.2 activator functions appear to be required to form a functional beta-cell. In this study, we have created transgenic mouse lines to express the Nkx2.2-repressor derivative in the mature beta-cell in the presence of endogenous Nkx2.2. The transgenic mice were assessed for beta-cell function, overall islet structure, and expression of beta-cell-specific markers. Using this transgenic approach, we have determined that the Nkx2.2-repressor derivative disrupts endogenous Nkx2.2 expression in adult mice and causes downregulation of the mature beta-cell factors, MafA and Glut2. Consistently, the Nkx2.2-repressor mice display reduced insulin gene expression and pancreatic insulin content and impaired insulin secretion. At weaning, the male Nkx2.2-repressor mice are overtly diabetic and all Nkx2.2-repressor transgenic mice exhibit glucose intolerance. Furthermore, the loss of beta-cell function in the Nkx2.2-repressor transgenic mice is associated with disrupted islet architecture. These studies indicate a previously undiscovered role for Nkx2.2 in the maintenance of mature beta-cell function and the formation of normal islet structure.
Figures

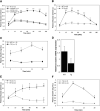

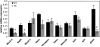
 ) mice (n = 4 each; islets were pooled from two mice for each sample). Nkx2.2, MafA, and Glut2 are significantly decreased in Nkx2.2-repressor islets. *P < 0.005. All other genes are not significantly changed. Error bars represent SEM of four individual samples for each genotype.
) mice (n = 4 each; islets were pooled from two mice for each sample). Nkx2.2, MafA, and Glut2 are significantly decreased in Nkx2.2-repressor islets. *P < 0.005. All other genes are not significantly changed. Error bars represent SEM of four individual samples for each genotype.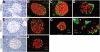

References
-
- Servitja JM, Ferrer J. Transcriptional networks controlling pancreatic development and β-cell function. Diabetologia. 2004;47:597–613. - PubMed
-
- Stoffers DA, Zinkin NT, Stanojevic V, Clarke WL, Habener JF. Pancreatic agenesis attributable to a single nucleotide deletion in the human IPF1 gene coding sequence. Nat Genet. 1997;15:106–110. - PubMed
-
- Malecki MT, Jhala US, Antonellis A, Fields L, Doria A, Orban T, Saad M, Warram JH, Montminy M, Krolewski AS. Mutations in NEUROD1 are associated with the development of type 2 diabetes mellitus. Nat Genet. 1999;23:323–328. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

