Persistent sodium currents participate in fictive locomotion generation in neonatal mouse spinal cord
- PMID: 17460064
- PMCID: PMC6673000
- DOI: 10.1523/JNEUROSCI.0124-07.2007
Persistent sodium currents participate in fictive locomotion generation in neonatal mouse spinal cord
Abstract
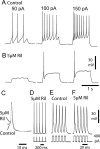
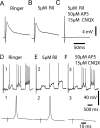
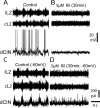
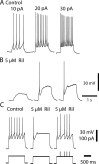
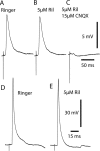
The persistent sodium current (I(Na(P))) has been implicated in the regulation of synaptic integration, intrinsic membrane properties, and rhythm generation in many types of neurons. We characterized I(Na(P)) in commissural interneurons (CINs) in the neonatal (postnatal days 0-3) mouse spinal cord; it is activated at subthreshold potentials, inactivates slowly, and can be blocked by low concentrations of riluzole. The role of I(Na(P)) in locomotor pattern generation was examined by applying riluzole during fictive locomotion induced by NMDA, serotonin, and dopamine or by stimulation of the cauda equina. Blockade of I(Na(P)) has marginal effects on the locomotion frequency but progressively weakens the rhythmic firing and locomotor-related membrane oscillation of CINs and motoneurons (MNs) and the locomotor-like bursts in ventral roots, until the motor pattern ceases. Riluzole directly affects the intrinsic firing properties of CINs and MNs, reducing their ability to fire repetitively during tonic depolarizations and raising their spike threshold. At the same time, riluzole has little effects on the strength of spike-evoked synaptic transmission onto CINs and MNs. Our results suggest that I(Na(P)) is essential for the generation of the locomotor pattern and acts in part by regulating the frequency of interneuron firing in the central pattern generator for locomotion.
Figures
Similar articles
-
Persistent sodium current contributes to induced voltage oscillations in locomotor-related hb9 interneurons in the mouse spinal cord.J Neurophysiol. 2008 Oct;100(4):2254-64. doi: 10.1152/jn.90437.2008. Epub 2008 Jul 30. J Neurophysiol. 2008. PMID: 18667543 Free PMC article.
-
Contribution of persistent sodium current to locomotor pattern generation in neonatal rats.J Neurophysiol. 2007 Aug;98(2):613-28. doi: 10.1152/jn.00316.2007. Epub 2007 Jun 13. J Neurophysiol. 2007. PMID: 17567773
-
Crossed rhythmic synaptic input to motoneurons during selective activation of the contralateral spinal locomotor network.J Neurosci. 1997 Dec 15;17(24):9433-47. doi: 10.1523/JNEUROSCI.17-24-09433.1997. J Neurosci. 1997. PMID: 9390999 Free PMC article.
-
Locomotor-like activity generated by the neonatal mouse spinal cord.Brain Res Brain Res Rev. 2002 Oct;40(1-3):141-51. doi: 10.1016/s0165-0173(02)00197-2. Brain Res Brain Res Rev. 2002. PMID: 12589913 Review.
-
Modeling the mammalian locomotor CPG: insights from mistakes and perturbations.Prog Brain Res. 2007;165:235-53. doi: 10.1016/S0079-6123(06)65015-2. Prog Brain Res. 2007. PMID: 17925250 Free PMC article. Review.
Cited by
-
Serotonergic Modulation of Persistent Inward Currents in Serotonergic Neurons of Medulla in ePet-EYFP Mice.Front Neural Circuits. 2021 Apr 6;15:657445. doi: 10.3389/fncir.2021.657445. eCollection 2021. Front Neural Circuits. 2021. PMID: 33889077 Free PMC article.
-
Prax330 reduces persistent and resurgent sodium channel currents and neuronal hyperexcitability of subiculum neurons in a mouse model of SCN8A epileptic encephalopathy.Neuropharmacology. 2019 Nov 1;158:107699. doi: 10.1016/j.neuropharm.2019.107699. Epub 2019 Jul 3. Neuropharmacology. 2019. PMID: 31278928 Free PMC article.
-
The persistent sodium current generates pacemaker activities in the central pattern generator for locomotion and regulates the locomotor rhythm.J Neurosci. 2008 Aug 20;28(34):8577-89. doi: 10.1523/JNEUROSCI.1437-08.2008. J Neurosci. 2008. PMID: 18716217 Free PMC article.
-
Molecular Mechanisms Underlying Cell Death in Spinal Networks in Relation to Locomotor Activity After Acute Injury in vitro.Front Cell Neurosci. 2011 Jun 17;5:9. doi: 10.3389/fncel.2011.00009. eCollection 2011. Front Cell Neurosci. 2011. PMID: 21734866 Free PMC article.
-
Control of oscillation periods and phase durations in half-center central pattern generators: a comparative mechanistic analysis.J Comput Neurosci. 2009 Aug;27(1):3-36. doi: 10.1007/s10827-008-0124-4. Epub 2009 Jan 6. J Comput Neurosci. 2009. PMID: 19130197 Free PMC article.
References
-
- Ahn HS, Choi JS, Choi BH, Kim MJ, Rhie DJ, Yoon SH, Jo YH, Kim MS, Sung KW, Hahn SJ. Inhibition of the cloned delayed rectifier K+ channels, Kv1.5 and Kv3.1, by riluzole. Neuroscience. 2005;133:1007–1019. - PubMed
-
- Albo F, Pieri M, Zona C. Modulation of AMPA receptors in spinal motor neurons by the neuroprotective agent riluzole. J Neurosci Res. 2004;78:200–207. - PubMed
-
- Buschges A, Wikstrom MA, Grillner S, El Manira A. Roles of high-voltage-activated calcium channel subtypes in a vertebrate spinal locomotor network. J Neurophysiol. 2000;84:2758–2766. - PubMed
-
- Butt SJ, Kiehn O. Functional identification of interneurons responsible for left-right coordination of hindlimbs in mammals. Neuron. 2003;38:953–963. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
