An improved high throughput protein-protein interaction assay for nuclear hormone receptors
- PMID: 17464356
- PMCID: PMC1853068
- DOI: 10.1621/nrs.05002
An improved high throughput protein-protein interaction assay for nuclear hormone receptors
Abstract
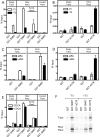
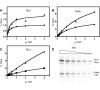
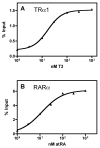
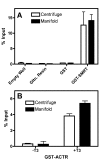
The Glutathione-S-Transferase (GST) "pulldown" assay has been used extensively to assay protein interactions in vitro. This methodology has been especially useful for investigating the interactions of nuclear hormone receptors with a wide variety of their interacting partners and coregulatory proteins. Unfortunately, the original GST-pulldown technique relies on multiple binding, washing and elution steps performed in individual microfuge tubes, and requires repeated centrifugation, aspiration, and suspension steps. This type of batch processing creates a significant liquid handling bottleneck, limiting the number of sample points that can be incorporated into one experiment and producing inherently less efficient washing and elution than would a flow-through methodology. In this manuscript, we describe the adaptation of this GST-pulldown assay to a 96-well filter plate format. The use of a multi-well filter plate makes it possible to assay more samples in significantly less time using less reagents and more efficient sample processing than does the traditional single tube assay.
Figures
Similar articles
-
High throughput analysis of nuclear receptor-cofactor interactions.Methods Mol Biol. 2009;505:157-69. doi: 10.1007/978-1-60327-575-0_9. Methods Mol Biol. 2009. PMID: 19117144
-
Study of G-protein-coupled receptor-protein interactions using gel overlay assays and glutathione-S-transferase-fusion protein pull-downs.Methods Mol Biol. 2004;259:371-8. doi: 10.1385/1-59259-754-8:371. Methods Mol Biol. 2004. PMID: 15250505
-
Glutathione-S-transferase-fusion based assays for studying protein-protein interactions.Methods Mol Biol. 2004;261:175-86. doi: 10.1385/1-59259-762-9:175. Methods Mol Biol. 2004. PMID: 15064458 Review.
-
Glutathione-S-transferase-green fluorescent protein fusion protein reveals slow dissociation from high site density beads and measures free GSH.Cytometry A. 2006 May;69(5):326-34. doi: 10.1002/cyto.a.20259. Cytometry A. 2006. PMID: 16604533
-
Protein-protein interactions identified by pull-down experiments and mass spectrometry.Curr Protoc Cell Biol. 2004 May;Chapter 17:Unit 17.5. doi: 10.1002/0471143030.cb1705s22. Curr Protoc Cell Biol. 2004. PMID: 18228443 Review.
Cited by
-
Alternative mRNA splicing of corepressors generates variants that play opposing roles in adipocyte differentiation.J Biol Chem. 2011 Dec 30;286(52):44988-99. doi: 10.1074/jbc.M111.291625. Epub 2011 Nov 7. J Biol Chem. 2011. PMID: 22065574 Free PMC article.
-
Nuclear receptor-coregulator interaction profiling identifies TRIP3 as a novel peroxisome proliferator-activated receptor gamma cofactor.Mol Cell Proteomics. 2009 Oct;8(10):2212-26. doi: 10.1074/mcp.M900209-MCP200. Epub 2009 Jul 10. Mol Cell Proteomics. 2009. PMID: 19596656 Free PMC article.
-
Thyroid hormone receptor mutations found in renal clear cell carcinomas alter corepressor release and reveal helix 12 as key determinant of corepressor specificity.Mol Endocrinol. 2009 Aug;23(8):1183-92. doi: 10.1210/me.2009-0126. Epub 2009 Apr 30. Mol Endocrinol. 2009. PMID: 19407221 Free PMC article.
-
Proteomics analysis of the estrogen receptor alpha receptosome.Mol Cell Proteomics. 2010 Jul;9(7):1411-22. doi: 10.1074/mcp.M900457-MCP200. Epub 2010 Mar 27. Mol Cell Proteomics. 2010. PMID: 20348541 Free PMC article.
-
Deciphering Mode of Action of Functionally Important Regions in the Intrinsically Disordered Paxillin (Residues 1-313) Using Its Interaction with FAT (Focal Adhesion Targeting Domain of Focal Adhesion Kinase).PLoS One. 2016 Feb 29;11(2):e0150153. doi: 10.1371/journal.pone.0150153. eCollection 2016. PLoS One. 2016. PMID: 26928467 Free PMC article.
References
-
- Apriletti J. W., Ribeiro R. C., Wagner R. L., Feng W., Webb P., Kushner P. J., West B. L., Nilsson S., Scanlan T. S., Fletterick R. J., Baxter J. D. Molecular and structural biology of thyroid hormone receptors. Clin Exp Pharmacol Physiol Suppl. 1998;25:S2–11. - PubMed
-
- Beato M., Klug J. Steroid hormone receptors: an update. Hum Reprod Update. 2000;6:225–36. - PubMed
-
- Bedouelle H., Duplay P. Production in Escherichia coli and one-step purification of bifunctional hybrid proteins which bind maltose. Export of the Klenow polymerase into the periplasmic space. Eur J Biochem. 1988;171:541–9. - PubMed
-
- Chambon P. A decade of molecular biology of retinoic acid receptors. Faseb J. 1996;10:940–54. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

