Phase I and correlative biology study of cilengitide in patients with recurrent malignant glioma
- PMID: 17470857
- PMCID: PMC3811028
- DOI: 10.1200/JCO.2006.06.6514
Phase I and correlative biology study of cilengitide in patients with recurrent malignant glioma
Abstract
Purpose: This multi-institutional phase I trial was designed to determine the maximum-tolerated dose (MTD) of cilengitide (EMD 121974) and to evaluate the use of perfusion magnetic resonance imaging (MRI) in patients with recurrent malignant glioma.
Patients and methods: Patients received cilengitide twice weekly on a continuous basis. A treatment cycle was defined as 4 weeks. Treatment-related dose-limiting toxicity (DLT) was defined as any grade 3 or 4 nonhematologic toxicity or grade 4 hematologic toxicity of any duration.
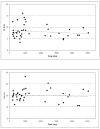
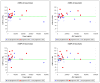
Results: A total of 51 patients were enrolled in cohorts of six patients to doses of 120, 240, 360, 480, 600, 1,200, 1,800, and 2,400 mg/m2 administered as a twice weekly intravenous infusion. Three patients progressed early and were inevaluable for toxicity assessment. The DLTs observed were one thrombosis (120 mg/m2), one grade 4 joint and bone pain (480 mg/m2), one thrombocytopenia (600 mg/m2) and one anorexia, hypoglycemia, and hyponatremia (800 mg/m2). The MTD was not reached. Two patients demonstrated complete response, three patients had partial response, and four patients had stable disease. Perfusion MRI revealed a significant relationship between the change in tumor relative cerebral blood flow (rCBF) from baseline and area under the plasma concentration versus time curve after 16 weeks of therapy.
Conclusion: Cilengitide is well tolerated to doses of 2,400 mg/m2, durable complete and partial responses were seen in this phase I study, and clinical response appears related to rCBF changes.
Figures

Comment in
-
Integrin inhibitors reaching the clinic.J Clin Oncol. 2007 May 1;25(13):1637-8. doi: 10.1200/JCO.2006.09.8376. J Clin Oncol. 2007. PMID: 17470853 No abstract available.
References
-
- Published by the Central Brain Tumor Registry of the United States CBTRUS: Statistical Report: Primary Brain Tumors in the United States 1997-2001. 2004
-
- Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352:987–996. - PubMed
-
- Tatter SB. Recurrent malignant glioma in adults. Curr Treat Options Oncol. 2002;3:509–524. - PubMed
-
- Libermann TA, Nusbaum HR, Razon N, et al. Amplification, enhanced expression and possible rearrangement of EGF receptor gene in primary human brain tumors of glial origin. Nature. 1985;313:144–147. - PubMed
-
- Watanabe K, Tachibana O, Sata K, et al. Overexpression of the EGF receptor and p53 mutations are mutually exclusive in the evolution of primary and secondary glioblastomas. Brain Pathol. 1996;6:217–223. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

