Pulmonary endothelial cell barrier enhancement by FTY720 does not require the S1P1 receptor
- PMID: 17475445
- PMCID: PMC2682440
- DOI: 10.1016/j.cellsig.2007.03.011
Pulmonary endothelial cell barrier enhancement by FTY720 does not require the S1P1 receptor
Abstract
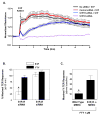
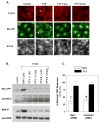
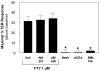
Novel therapeutic strategies are needed to reverse the loss of endothelial cell (EC) barrier integrity that occurs during inflammatory disease states such as acute lung injury. We previously demonstrated potent EC barrier augmentation in vivo and in vitro by the platelet-derived phospholipid, sphingosine 1-phosphate (S1P) via ligation of the S1P1 receptor. The S1P analogue, FTY720, similarly exerts barrier-protective vascular effects via presumed S1P1 receptor ligation. We examined the role of the S1P1 receptor in sphingolipid-mediated human lung EC barrier enhancement. Both S1P and FTY-induced sustained, dose-dependent barrier enhancement, reflected by increases in transendothelial electrical resistance (TER), which was abolished by pertussis toxin indicating Gi-coupled receptor activation. FTY-mediated increases in TER exhibited significantly delayed onset and intensity relative to the S1P response. Reduction of S1P1R expression (via siRNA) attenuated S1P-induced TER elevations whereas the TER response to FTY was unaffected. Both S1P and FTY rapidly (within 5 min) induced S1P1R accumulation in membrane lipid rafts, but only S1P stimulated S1P1R phosphorylation on threonine residues. Inhibition of PI3 kinase activity attenuated S1P-mediated TER increases but failed to alter FTY-induced TER elevation. Finally, S1P, but not FTY, induced significant myosin light chain phosphorylation and dramatic actin cytoskeletal rearrangement whereas reduced expression of the cytoskeletal effectors, Rac1 and cortactin (via siRNA), attenuated S1P-, but not FTY-induced TER elevations. These results mechanistically characterize pulmonary vascular barrier regulation by FTY720, suggesting a novel barrier-enhancing pathway for modulating vascular permeability.
Figures


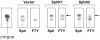


References
-
- Dudek SM, Garcia JGJ. Appl Physiol. 2001;91(4):1487–1500. - PubMed
-
- Shikata Y, Birukov KG, Garcia JG. J Appl Physiol. 2003;94(3):1193–1203. - PubMed
-
- Dudek SM, Jacobson JR, Chiang ET, Birukov KG, Wang P, Zhan X, Garcia JG. J Biol Chem. 2004;279(23):24692–24700. - PubMed
-
- McVerry BJ, Peng X, Hassoun PM, Sammani S, Simon BA, Garcia JG. Am J Respir Crit Care Med. 2004;170(9):987–993. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

