Expression of the LIM-homeodomain protein Isl1 in the developing and mature mouse retina
- PMID: 17480014
- PMCID: PMC2950632
- DOI: 10.1002/cne.21390
Expression of the LIM-homeodomain protein Isl1 in the developing and mature mouse retina
Abstract
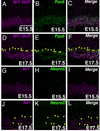
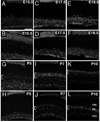
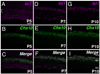
The mammalian retina is comprised of six major neuronal cell types and is subdivided into more morphological and physiological subtypes. The transcriptional machinery underlying these subtype fate choices is largely unknown. The LIM-homeodomain protein, Isl1, plays an essential role in central nervous system (CNS) differentiation but its relationship to retinal neurogenesis remains unknown. We report here its dynamic spatiotemporal expression in the mouse retina. Among bipolar interneurons, Isl1 expression commences at postnatal day (P)5 and is later restricted to ON-bipolar cells. The intensity of Isl1 expression is found to segregate the pool of ON-bipolar cells into rod and ON-cone bipolar cells with higher expression in rod bipolar cells. As bipolar cell development proceeds from P5-10 the colocalization of Isl1 and the pan-bipolar cell marker Chx10 reveals the organization of ON-center bipolar cell nuclei to the upper portion of the inner nuclear layer. Further, whereas Isl1 is predominantly a ganglion cell marker prior to embryonic day (E)15.5, at E15.5 and later its expression in nonganglion cells expands. We demonstrate that these Isl1-positive, nonganglion cells acquire the expression of amacrine cell markers embryonically, likely representing nascent cholinergic amacrine cells. Taken together, Isl1 is expressed during the maturation of and is later maintained in retinal ganglion cells and subtypes of amacrine and bipolar cells where it may function in the maintenance of these cells into adulthood.
Figures







References
-
- Beleky-Adams T, Tomarev S, Li HS, Ploder L, McInnes RR, Sundin O, Adler R. Pax-6, Prox 1, and Chx10 homeobox gene expression correlates with phenotypic fate of retinal precursor cells. Invest Ophthalmol Vis Sci. 1997;38:1293–1303. - PubMed
-
- Bramblett DE, Pennesi ME, Wu SM, Tsai MJ. The transcription factor Bhlhb4 is required for rod bipolar cell maturation. Neuron. 2004;43:779–793. - PubMed
-
- Brown SP, Masland RH. Costratification of a population of bipolar cells with the direction-selective circuitry of the rabbit retina. J Comp Neurol. 1999;408:97–106. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

