Lipopolysaccharide induces delayed FosB/DeltaFosB immunostaining within the mouse extended amygdala, hippocampus and hypothalamus, that parallel the expression of depressive-like behavior
- PMID: 17482371
- PMCID: PMC1978247
- DOI: 10.1016/j.psyneuen.2007.03.005
Lipopolysaccharide induces delayed FosB/DeltaFosB immunostaining within the mouse extended amygdala, hippocampus and hypothalamus, that parallel the expression of depressive-like behavior
Abstract
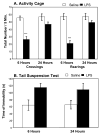
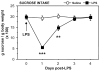
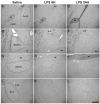
Proinflammatory cytokines induce both sickness behavior and depression, but their respective neurobiological correlates are still poorly understood. The aim of the present study was therefore to identify in mice the neural substrates of sickness and depressive-like behavior induced by lipopolysaccharide (LPS, 830 microg/kg, intraperitoneal). LPS-induced depressive-like behavior was dissociated from LPS-induced sickness by testing mice either at 6 h (at which time sickness was expected to be maximal) or at 24 h post-LPS (at which time sickness was expected to be minimal and not to bias the measurement of depressive-like behavior). Concurrently, the expression of acute and chronic cellular reactivity markers (c-Fos and FosB/DeltaFosB, respectively) was mapped by immunohistochemistry at these two time points. In comparison to saline, LPS decreased motor activity in a new cage at 6 h but not at 24 h. In contrast, the duration of immobility in the tail suspension test was increased at both 6 and 24 h. This dissociation between decreased motor activity and depressive-like behavior was confirmed at 24 h post-LPS in the forced swim test. LPS also decreased sucrose consumption at 24 and 48 h, despite normal food and water consumption by that time. At 24 h post-LPS, LPS-induced depressive-like behavior was associated with a delayed cellular activity (as assessed by FosB/DeltaFosB immunostaining) in specific brain structures, particularly within the extended amygdala, hippocampus and hypothalamus, whereas c-Fos labeling was markedly decreased by that time in all the brain areas at 6 h post-LPS. These results provide the first evidence in favor of a functional dissociation between the brain structures that underlie cytokine-induced sickness behavior and cytokine-induced depressive-like behavior, and provide important cues about the neuroanatomical brain circuits through which cytokines could have an impact on affect.
Figures



References
-
- Alheid GF. Extended amygdala and basal forebrain. Ann NY Acad Sci. 2003;985:185–205. - PubMed
-
- Alheid GF, Heimer L. New perspectives in basal forebrain organization of special relevance for neuropsychiatric disorders: the striatopallidal, amygdaloid, and corticopetal components of substantia innominata. Neuroscience. 1988;27:1–39. - PubMed
-
- Alheid GF, Heimer L. Theories of basal forebrain organization and the “emotional motor system”. Prog Brain Res. 1996;107:461–484. - PubMed
-
- Allan SM, Rothwell NJ. Cytokines and acute neurodegeneration. Nat Rev Neurosci. 2001;2:734–744. - PubMed
-
- Anisman H, Kokkinidis L, Merali Z. Further evidence for the depressive effects of cytokines: anhedonia and neurochemical changes. Brain Behav Immun. 2002;16:544–556. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

