The lysophosphatidic acid type 2 receptor is required for protection against radiation-induced intestinal injury
- PMID: 17484878
- PMCID: PMC3446791
- DOI: 10.1053/j.gastro.2007.03.038
The lysophosphatidic acid type 2 receptor is required for protection against radiation-induced intestinal injury
Abstract
Background & aims: We recently identified lysophosphatidic acid (LPA) as a potent antiapoptotic agent for the intestinal epithelium. The objective of the present study was to evaluate the effect of octadecenyl thiophosphate (OTP), a novel rationally designed, metabolically stabilized LPA mimic, on radiation-induced apoptosis of intestinal epithelial cells in vitro and in vivo.
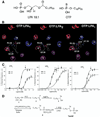
Methods: The receptors and signaling pathways activated by OTP were examined in IEC-6 and RH7777 cell lines and wild-type and LPA(1) and LPA(2) knockout mice exposed to different apoptotic stimuli.
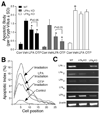
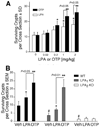
Results: OTP was more efficacious than LPA in reducing gamma irradiation-, camptothecin-, or tumor necrosis factor alpha/cycloheximide-induced apoptosis and caspase-3-8, and caspase-9 activity in the IEC-6 cell line. In RH7777 cells lacking LPA receptors, OTP selectively protected LPA(2) but not LPA(1) and LPA(3) transfectants. In C57BL/6 and LPA(1) knockout mice exposed to 15 Gy gamma irradiation, orally applied OTP reduced the number of apoptotic bodies and activated caspase-3-positive cells but was ineffective in LPA(2) knockout mice. OTP, with higher efficacy than LPA, enhanced intestinal crypt survival in C57BL/6 mice but was without any effect in LPA(2) knockout mice. Intraperitoneally administered OTP reduced death caused by lethal dose (LD)(100/30) radiation by 50%.
Conclusions: Our data indicate that OTP is a highly effective antiapoptotic agent that engages similar prosurvival pathways to LPA through the LPA(2) receptor subtype.
Figures







References
-
- Potten CS. Radiation, the ideal cytotoxic agent for studying the cell biology of tissues such as the small intestine. Radiat Res. 2004;161:123–136. - PubMed
-
- Tigyi G, Parrill AL. Molecular mechanisms of lysophosphatidic acid action. Prog Lipid Res. 2003;42:498–526. - PubMed
-
- Moolenaar WH, van Meeteren LA, Giepmans BN. The ins and outs of lysophosphatidic acid signaling. Bioessays. 2004;26:870–881. - PubMed
-
- Ishii I, Fukushima N, Ye X, Chun J. Lysophospholipid receptors: signaling and biology. Annu Rev Biochem. 2004;73:321–354. - PubMed
-
- Noguchi K, Ishii S, Shimizu T. Identification of p2y9/GPR23 as a novel G protein-coupled receptor for lysophosphatidic acid, structurally distant from the Edg family. J Biol Chem. 2003;278:25600–25606. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

