CCR7 ligands control basal T cell motility within lymph node slices in a phosphoinositide 3-kinase-independent manner
- PMID: 17485513
- PMCID: PMC2118589
- DOI: 10.1084/jem.20062079
CCR7 ligands control basal T cell motility within lymph node slices in a phosphoinositide 3-kinase-independent manner
Abstract
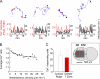
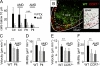
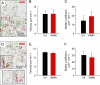
The molecular mechanisms responsible for the sustained basal motility of T cells within lymph nodes (LNs) remain elusive. To study T cell motility in a LN environment, we have developed a new experimental system based on slices of LNs that allows the assessment of T cell trafficking after adoptive transfer or direct addition of T cells to the slice. Using this experimental system, we show that T cell motility is highly sensitive to pertussis toxin and strongly depends on CCR7 and its ligands. Our results also demonstrate that, despite its established role in myeloid cell locomotion, phosphoinositide 3-kinase (PI3K) activity does not contribute to the exploratory behavior of the T lymphocytes within LN slices. Likewise, although PI3K activation is detectable in chemokine-treated T cells, PI3K plays only a minor role in T cell polarization and migration in vitro. Collectively, our results suggest that the common amplification system that, in other cells, facilitates large phosphatidylinositol 3,4,5-trisphosphate increases at the plasma membrane is absent in T cells. We conclude that T cell motility within LNs is not an intrinsic property of T lymphocytes but is driven in a PI3K-independent manner by the lymphoid chemokine-rich environment.
Figures


References
-
- Cyster, J.G. 2005. Chemokines, sphingosine-1-phosphate, and cell migration in secondary lymphoid organs. Annu. Rev. Immunol. 23:127–159. - PubMed
-
- Nakano, H., S. Mori, H. Yonekawa, H. Nariuchi, A. Matsuzawa, and T. Kakiuchi. 1998. A novel mutant gene involved in T-lymphocyte-specific homing into peripheral lymphoid organs on mouse chromosome 4. Blood. 91:2886–2895. - PubMed
-
- Forster, R., A. Schubel, D. Breitfeld, E. Kremmer, I. Renner-Muller, E. Wolf, and M. Lipp. 1999. CCR7 coordinates the primary immune response by establishing functional microenvironments in secondary lymphoid organs. Cell. 99:23–33. - PubMed
-
- Bousso, P., and E. Robey. 2003. Dynamics of CD8+ T cell priming by dendritic cells in intact lymph nodes. Nat. Immunol. 4:579–585. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

