Non-genomic action of resveratrol on androgen and oestrogen receptors in prostate cancer: modulation of the phosphoinositide 3-kinase pathway
- PMID: 17486135
- PMCID: PMC2359939
- DOI: 10.1038/sj.bjc.6603755
Non-genomic action of resveratrol on androgen and oestrogen receptors in prostate cancer: modulation of the phosphoinositide 3-kinase pathway
Abstract
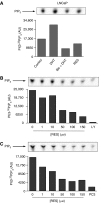
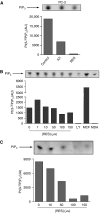
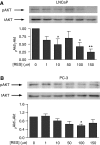
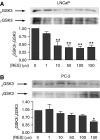
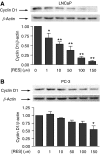
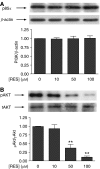
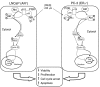
Prostate cancer represents a major concern in human oncology and the phytoalexin resveratrol (RES) inhibits growth and proliferation of prostate cancer cells through the induction of apoptosis. In addition, previous data indicate that in oestrogen-responsive human breast cancer cells, RES induces apoptosis by inhibition of the phosphoinositide-3-kinase (PI3K) pathway. Here, using androgen receptor (AR)-positive LNCaP and oestrogen receptor alpha (ERalpha)-expressing PC-3 prostate tumour cells, we have analysed whether the antiproliferative activity of RES takes place by inhibition of the AR- or ERalpha-dependent PI3K pathway. Although RES treatment (up to 150 microM) decreased AR and ERalpha protein levels, it did not affect AR and ERalpha interaction with p85-PI3K. Immunoprecipitation and kinase assays showed that RES inhibited AR- and ERalpha-dependent PI3K activities in LNCaP and PC-3, respectively. Consistently, lower PI3K activities correlated with decreased phosphorylation of downstream targets protein kinase B/AKT (PKB/AKT) and glycogen synthase kinase-3 (GSK-3). GSK-3 dephosphorylation could be responsible for the decreased cyclin D1 levels observed in both cell lines. Importantly, RES markedly decreased PKB/AKT phosphorylation in primary cultures from human prostate tumours, suggesting that the mechanism proposed here could take place in vivo. Thus, RES could have antitumoral activity in androgen-sensitive and androgen-non-sensitive human prostate tumours by inhibiting survival pathways such as that mediated by PI3K.
Figures

Similar articles
-
Resveratrol modulates the phosphoinositide 3-kinase pathway through an estrogen receptor alpha-dependent mechanism: relevance in cell proliferation.Int J Cancer. 2004 Mar 20;109(2):167-73. doi: 10.1002/ijc.11720. Int J Cancer. 2004. PMID: 14750165
-
Signal transduction pathways in androgen-dependent and -independent prostate cancer cell proliferation.Endocr Relat Cancer. 2005 Mar;12(1):119-34. doi: 10.1677/erc.1.00835. Endocr Relat Cancer. 2005. PMID: 15788644
-
PI3K-AKT-mTOR pathway is dominant over androgen receptor signaling in prostate cancer cells.Cell Oncol. 2010;32(1-2):11-27. doi: 10.3233/CLO-2009-0487. Cell Oncol. 2010. PMID: 20203370 Free PMC article.
-
Cross-talk between the androgen receptor and the phosphatidylinositol 3-kinase/Akt pathway in prostate cancer.Curr Cancer Drug Targets. 2007 Sep;7(6):591-604. doi: 10.2174/156800907781662248. Curr Cancer Drug Targets. 2007. PMID: 17896924 Review.
-
Mechanisms of prostate cancer cell survival after inhibition of AR expression.J Cell Biochem. 2009 Feb 15;106(3):363-71. doi: 10.1002/jcb.22022. J Cell Biochem. 2009. PMID: 19115258 Review.
Cited by
-
Resveratrol enhances polyubiquitination-mediated ARV7 degradation in prostate cancer cells.Oncotarget. 2017 May 19;8(33):54683-54693. doi: 10.18632/oncotarget.18003. eCollection 2017 Aug 15. Oncotarget. 2017. PMID: 28903374 Free PMC article.
-
The Effects of Resveratrol on Prostate Cancer through Targeting the Tumor Microenvironment.J Xenobiot. 2021 Feb 1;11(1):16-32. doi: 10.3390/jox11010002. J Xenobiot. 2021. PMID: 33535458 Free PMC article. Review.
-
Molecular Mechanisms and Bioavailability of Polyphenols in Prostate Cancer.Int J Mol Sci. 2019 Mar 1;20(5):1062. doi: 10.3390/ijms20051062. Int J Mol Sci. 2019. PMID: 30823649 Free PMC article. Review.
-
Phytochemicals and PI3K Inhibitors in Cancer-An Insight.Front Pharmacol. 2017 Dec 14;8:916. doi: 10.3389/fphar.2017.00916. eCollection 2017. Front Pharmacol. 2017. PMID: 29311925 Free PMC article. Review.
-
Targeting GSK3 and Associated Signaling Pathways Involved in Cancer.Cells. 2020 Apr 30;9(5):1110. doi: 10.3390/cells9051110. Cells. 2020. PMID: 32365809 Free PMC article. Review.
References
-
- Ahmad N, Adhami VM, Afaq F, Feyes DK, Mukhtar H (2001) Resveratrol causes WAF-1/p21-mediated G(1)-phase arrest of cell cycle and induction of apoptosis in human epidermoid carcinoma A431 cells. Clin Cancer Res 7: 1466–1473 - PubMed
-
- Alessi DR, James SR, Downes CP, Holmes AB, Gaffney PR, Reese CB, Cohen P (1997) Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Balpha. Curr Biol 7: 261–269 - PubMed
-
- Aziz MH, Nihal M, Fu VX, Jarrard DF, Ahmad N (2006) Resveratrol-caused apoptosis of human prostate carcinoma LNCaP cells is mediated via modulation of phosphatidylinositol 3′-kinase/Akt pathway and Bcl-2 family proteins. Mol Cancer Ther 5: 1335–1341 - PubMed
-
- Banerjee S, Bueso-Ramos C, Aggarwal BB (2002) Suppression of 7,12-dimethylbenz(a)anthracene-induced mammary carcinogenesis in rats by resveratrol: role of nuclear factor-kappaB, cyclooxygenase 2, and matrix metalloprotease 9. Cancer Res 62: 4945–4954 - PubMed
-
- Basly JP, Marre-Fournier F, Le Bail JC, Habrioux G, Chulia AJ (2000) Estrogenic/antiestrogenic and scavenging properties of (E)- and (Z)-resveratrol. Life Sci 66: 769–777 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

