Prenatal stress enhances responsiveness to cocaine
- PMID: 17487224
- PMCID: PMC2747653
- DOI: 10.1038/sj.npp.1301447
Prenatal stress enhances responsiveness to cocaine
Abstract
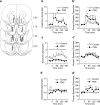
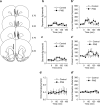
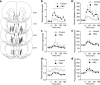
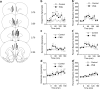
Early environmental events have profound influences on a wide range of adult behavior. In the current study, we assessed the influence of maternal stress during gestation on psychostimulant and neurochemical responsiveness to cocaine, cocaine self-administration, and reinstatement of cocaine-seeking in adult offspring. Pregnant, female Sprague-Dawley rats were subjected to either no treatment or to restraint stress three times per day for the last 7 days of gestation and cocaine-related behavior was assessed in offspring at 10 weeks of age. Relative to controls, a noncontingent cocaine injection elevated locomotor activity as well as nucleus accumbens levels of extracellular dopamine and glutamate to a greater extent in both cocaine-naive and cocaine-experienced prenatal stress (PNS) rats and elevated prefrontal cortex dopamine in cocaine-experienced PNS rats. To assess the impact of PNS on cocaine addiction-related behavior, rats were trained to lever press for intravenous (i.v.) infusions of cocaine (0.25, 0.5, or 1 mg/kg/infusion), with each infusion paired with a light+tone-conditioned stimulus. Lever-pressing was extinguished and cocaine-seeking reinstated by re-exposure to the conditioned cues or by intraperitoneal cocaine-priming injections (5 or 10 mg/kg). PNS elevated active lever responding both during extinction and cocaine-primed reinstatement, but not during self-administration or conditioned-cued reinstatement. PNS also did not alter intake during self-administration. These findings demonstrate that PNS produces enduring nervous system alterations that increase the psychomotor stimulant, motivational, and neurochemical responsiveness to noncontingent cocaine. Thus, early environmental factors contribute to an individual's initial responsiveness to cocaine and propensity to relapse to cocaine-seeking.
Figures


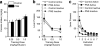
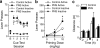
Similar articles
-
Cocaine administered into the medial prefrontal cortex reinstates cocaine-seeking behavior by increasing AMPA receptor-mediated glutamate transmission in the nucleus accumbens.J Neurosci. 2002 Apr 1;22(7):2916-25. doi: 10.1523/JNEUROSCI.22-07-02916.2002. J Neurosci. 2002. PMID: 11923456 Free PMC article.
-
Prefrontal glutamate release into the core of the nucleus accumbens mediates cocaine-induced reinstatement of drug-seeking behavior.J Neurosci. 2003 Apr 15;23(8):3531-7. doi: 10.1523/JNEUROSCI.23-08-03531.2003. J Neurosci. 2003. PMID: 12716962 Free PMC article.
-
Administration of the D2 dopamine receptor antagonist sulpiride into the shell, but not the core, of the nucleus accumbens attenuates cocaine priming-induced reinstatement of drug seeking.Neuropsychopharmacology. 2006 Jul;31(7):1452-61. doi: 10.1038/sj.npp.1300922. Epub 2005 Oct 5. Neuropsychopharmacology. 2006. PMID: 16205775
-
Contribution of early environmental stress to alcoholism vulnerability.Alcohol. 2009 Nov;43(7):547-54. doi: 10.1016/j.alcohol.2009.09.029. Alcohol. 2009. PMID: 19913199 Free PMC article. Review.
-
Addictive behaviour in experimental animals: prospects for translation.Philos Trans R Soc Lond B Biol Sci. 2018 Mar 19;373(1742):20170027. doi: 10.1098/rstb.2017.0027. Philos Trans R Soc Lond B Biol Sci. 2018. PMID: 29352026 Free PMC article. Review.
Cited by
-
Homer2 regulates alcohol and stress cross-sensitization.Addict Biol. 2016 May;21(3):613-33. doi: 10.1111/adb.12252. Epub 2015 Apr 27. Addict Biol. 2016. PMID: 25916683 Free PMC article.
-
Sex differences in salivary cortisol in response to acute stressors among healthy participants, in recreational or pathological gamblers, and in those with posttraumatic stress disorder.Horm Behav. 2010 Jan;57(1):35-45. doi: 10.1016/j.yhbeh.2009.06.003. Epub 2009 Jun 16. Horm Behav. 2010. PMID: 19538960 Free PMC article. Review.
-
Enduring, Sexually Dimorphic Impact of In Utero Exposure to Elevated Levels of Glucocorticoids on Midbrain Dopaminergic Populations.Brain Sci. 2016 Dec 30;7(1):5. doi: 10.3390/brainsci7010005. Brain Sci. 2016. PMID: 28042822 Free PMC article. Review.
-
Unravelling the Link Between Prenatal Stress, Dopamine and Substance Use Disorder.Neurotox Res. 2017 Jan;31(1):169-186. doi: 10.1007/s12640-016-9674-9. Epub 2016 Oct 24. Neurotox Res. 2017. PMID: 27778246 Review.
-
Stress and cocaine interact to modulate Arc/Arg3.1 expression in rat brain.Psychopharmacology (Berl). 2011 Nov;218(1):241-8. doi: 10.1007/s00213-011-2331-3. Epub 2011 May 18. Psychopharmacology (Berl). 2011. PMID: 21590283
References
-
- Alonso SJ, Navarro E, Rodriguez M. Permanent dopaminergic alterations in then accumbens after prenatal stress. Pharmacol Biochem Behav. 1994;49:353–358. - PubMed
-
- Anderson SM, Bari AA, Pierce RC. Administration of the D1-like dopamine receptor antagonist SCH-23390 into the medial nucleus accumbens shell attenuates cocaine priming-induced reinstatement of drug-seeking behavior in rats. Psychopharmacology. 2003;168:132–138. - PubMed
-
- Anderson SM, Pierce RC. Cocaine-induced alterations in dopamine receptor signaling: implications for reinforcement and reinstatement. Pharmacol Ther. 2005;106:389–403. - PubMed
-
- Anderson SM, Schmidt HD, Pierce RC. Administration of the D2 dopamine receptor antagonist sulpiride into the shell, but not the core, of the nucleus accumbens attenuates cocaine priming-induced reinstatement of drug seeking. Neuropsychopharmacology. 2006;31:1452–1461. - PubMed
-
- Bachtell RK, Whisler K, Karanian D, Self DW. Effects of intra-nucleus accumbens shell administration of dopamine agonists and antagonists on cocaine-taking and cocaine-seeking behaviors in the rat. Psychopharmacology. 2005;183:41–53. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

