N-glycan alterations are associated with drug resistance in human hepatocellular carcinoma
- PMID: 17488527
- PMCID: PMC1878497
- DOI: 10.1186/1476-4598-6-32
N-glycan alterations are associated with drug resistance in human hepatocellular carcinoma
Abstract
Background: Correlations of disease phenotypes with glycosylation changes have been analysed intensively in the tumor biology field. Glycoforms potentially associated with carcinogenesis, tumor progression and cancer metastasis have been identified. In cancer therapy, drug resistance is a severe problem, reducing therapeutic effect of drugs and adding to patient suffering. Although multiple mechanisms likely underlie resistance of cancer cells to anticancer drugs, including overexpression of transporters, the relationship of glycans to drug resistance is not well understood.
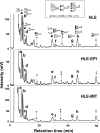
Results: We established epirubicin (EPI)--and mitoxantrone (MIT)--resistant cell lines (HLE-EPI and HLE-MIT) from the human hepatocellular carcinoma cell line (HLE). HLE-EPI and HLE-MIT overexpressed transporters MDR1/ABCB1 and BCRP/ABCG2, respectively. Here we compared the glycomics of HLE-EPI and HLE-MIT cells with the parental HLE line. Core fucosylated triantennary oligosaccharides were increased in the two resistant lines. We investigated mRNA levels of glycosyltransferases synthesizing this oligosaccharide, namely, N-acetylglucosaminyltransferase (GnT)-IVa, GnT-IVb and alpha1,6-fucosyltransferase (alpha1,6-FucT), and found that alpha1,6-FucT was particularly overexpressed in HLE-MIT cells. In HLE-EPI cells, GnT-IVa expression was decreased, while GnT-IVb was increased. Both GnT-IVs were downregulated in HLE-MIT cells. HLE-MIT cells also showed decreases in fucosylated tetraantennary oligosaccharide, the product of GnT-V. GnT-V expression was decreased in both lines, but particularly so in HLE-MIT cells. Thus both N-glycan and glycosyltransferase expression was altered as cells acquired tolerance, suggesting novel mechanisms of drug resistance.
Conclusion: N-glycan and glycosyltransferase expression in HLE-EPI and HLE-MIT were analysed and presented that glycans altered according with acquired tolerance. These results suggested novel mechanisms of drug resistance.
Figures




Similar articles
-
Expression of ABC transporters in human hepatocyte carcinoma cells with cross-resistance to epirubicin and mitoxantrone.Anticancer Res. 2006 Mar-Apr;26(2A):885-8. Anticancer Res. 2006. PMID: 16619483
-
Specific N-glycans of Hepatocellular Carcinoma Cell Surface and the Abnormal Increase of Core-α-1, 6-fucosylated Triantennary Glycan via N-acetylglucosaminyltransferases-IVa Regulation.Sci Rep. 2015 Nov 5;5:16007. doi: 10.1038/srep16007. Sci Rep. 2015. PMID: 26537865 Free PMC article.
-
Mammalian α-1,6-Fucosyltransferase (FUT8) Is the Sole Enzyme Responsible for the N-Acetylglucosaminyltransferase I-independent Core Fucosylation of High-mannose N-Glycans.J Biol Chem. 2016 May 20;291(21):11064-71. doi: 10.1074/jbc.M116.720789. Epub 2016 Mar 23. J Biol Chem. 2016. PMID: 27008861 Free PMC article.
-
Enzymes for N-Glycan Branching and Their Genetic and Nongenetic Regulation in Cancer.Biomolecules. 2016 Apr 28;6(2):25. doi: 10.3390/biom6020025. Biomolecules. 2016. PMID: 27136596 Free PMC article. Review.
-
Glycans and cancer: role of N-glycans in cancer biomarker, progression and metastasis, and therapeutics.Adv Cancer Res. 2015;126:11-51. doi: 10.1016/bs.acr.2014.11.001. Epub 2015 Feb 7. Adv Cancer Res. 2015. PMID: 25727145 Review.
Cited by
-
High expression of Lewis y antigen and CD44 is correlated with resistance to chemotherapy in epithelial ovarian cancers.PLoS One. 2013;8(2):e57250. doi: 10.1371/journal.pone.0057250. Epub 2013 Feb 28. PLoS One. 2013. PMID: 23468946 Free PMC article.
-
Xmrk, kras and myc transgenic zebrafish liver cancer models share molecular signatures with subsets of human hepatocellular carcinoma.PLoS One. 2014 Mar 14;9(3):e91179. doi: 10.1371/journal.pone.0091179. eCollection 2014. PLoS One. 2014. PMID: 24633177 Free PMC article.
-
Axl glycosylation mediates tumor cell proliferation, invasion and lymphatic metastasis in murine hepatocellular carcinoma.World J Gastroenterol. 2012 Oct 14;18(38):5369-76. doi: 10.3748/wjg.v18.i38.5369. World J Gastroenterol. 2012. PMID: 23082053 Free PMC article.
-
Swainsonine reduces 5-fluorouracil tolerance in the multistage resistance of colorectal cancer cell lines.Mol Cancer. 2007 Sep 21;6:58. doi: 10.1186/1476-4598-6-58. Mol Cancer. 2007. PMID: 17883871 Free PMC article.
-
Glycobiology of Cancer: Sugar Drives the Show.Medicines (Basel). 2022 May 24;9(6):34. doi: 10.3390/medicines9060034. Medicines (Basel). 2022. PMID: 35736247 Free PMC article.
References
-
- Yamashita K, Koide N, Endo T, Iwaki Y, Kobata A. Altered glycosylation of serum transferrin of patients with hepatocellular carcinoma. J Biol Chem. 1989;264:2415–2423. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

