Ih without Kir in adult rat retinal ganglion cells
- PMID: 17488978
- PMCID: PMC3241988
- DOI: 10.1152/jn.01241.2006
Ih without Kir in adult rat retinal ganglion cells
Abstract
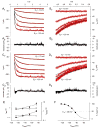
Antisera directed against hyperpolarization-activated mixed-cation ("I(h)") and K(+) ("K(ir)") channels bind to some somata in the ganglion cell layer of rat and rabbit retina. Additionally, the termination of hyperpolarizing current injections can trigger spikes in some cat retinal ganglion cells, suggesting a rebound depolarization arising from activation of I(h). However, patch-clamp studies showed that rat ganglion cells lack inward rectification or present an inwardly rectifying K(+) current. We therefore tested whether hyperpolarization activates I(h) in dissociated, adult rat retinal ganglion cell somata. We report here that, although we found no inward rectification in some cells, and a K(ir)-like current in a few cells, hyperpolarization activated I(h) in roughly 75% of the cells we recorded from in voltage clamp. We show that this current is blocked by Cs(+) or ZD7288 and only slightly reduced by Ba(2+), that the current amplitude and reversal potential are sensitive to extracellular Na(+) and K(+), and that we found no evidence of K(ir) in cells presenting I(h). In current clamp, injecting hyperpolarizing current induced a slowly relaxing membrane hyperpolarization that rebounded to a few action potentials when the hyperpolarizing current was stopped; both the membrane potential relaxation and rebound spikes were blocked by ZD7288. These results provide the first measurement of I(h) in mammalian retinal ganglion cells and indicate that the ion channels of rat retinal ganglion cells may vary in ways not expected from previous voltage and current recordings.
Figures



References
-
- Akamine T, Nishimura Y, Ito K, Uji Y, Yamamoto T. Effects of haloperidol on K+ currents in acutely isolated rat retinal ganglion cells. Invest Ophthalmol Vis Sci. 2002;43:1257–1261. - PubMed
-
- Akopian A, Witkovsky P. D2 dopamine receptor-mediated inhibition of a hyperpolarization-activated current in rod photoreceptors. J Neurophysiol. 1996;76:1828–1835. - PubMed
-
- Ames A, 3rd, Nesbett FB. In vitro retina as an experimental model of the central nervous system. J Neurochem. 1981;37:867–877. - PubMed
-
- Barres BA, Silverstein BE, Corey DP, Chun LL. Immunological, morphological, and electrophysiological variation among retinal ganglion cells purified by panning. Neuron. 1988;1:791–803. - PubMed
-
- Bayliss DA, Viana F, Bellingham MC, Berger AJ. Characteristics and postnatal development of a hyperpolarisation-activated inward current in rat hypoglossal motoneurones in vitro. J Neurophysiol. 1994;71:119–128. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

