Presynaptic AMPA and kainate receptors increase the size of GABAergic terminals and enhance GABA release
- PMID: 17493642
- PMCID: PMC2048687
- DOI: 10.1016/j.neuropharm.2007.03.010
Presynaptic AMPA and kainate receptors increase the size of GABAergic terminals and enhance GABA release
Abstract
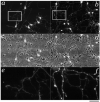
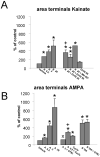
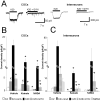
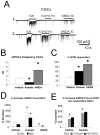
In the developing cerebellum, NMDA receptors promote the maturation of axonal terminals of inhibitory interneurons. We compared the effects of AMPA/kainate receptor agonists in cultured cerebellar cells from GAD65-eGFP mice. Both AMPA and kainate augmented granule cell survival without affecting interneurons. The action of kainate was blocked by an AMPA but not by a NMDA receptor antagonist, suggesting AMPA receptor involvement. AMPA and kainate increased the size of the GABAergic terminals and the action of kainate was insensitive to NMDA blockers. Whole cell recordings in granule neurons revealed that chronic treatment for 5 days with kainate as well as NMDA decreased AMPA receptor expression while interneuronal kainate receptors were depressed by kainate treatment. Acute kainate application increased mIPSCs frequency in both granule neurons and interneurons and this effect was only partially blocked by an AMPA receptor antagonist. In contrast to what was reported for NMDA, chronic treatment with kainate induced a significant decrease of the basal mIPSCs frequency but increased the acute action of kainate on mIPSCs. Direct recordings from presynaptic GABAergic terminals suggest that AMPA and kainate receptors are present in developing GABAergic terminals and their activation affects the size of GABAergic terminals and spontaneous GABA release.
Figures

Similar articles
-
Biphasic modulation of GABA release from stellate cells by glutamatergic receptor subtypes.J Neurophysiol. 2007 Jul;98(1):550-6. doi: 10.1152/jn.00352.2007. Epub 2007 May 30. J Neurophysiol. 2007. PMID: 17537903
-
NMDA receptors increase the size of GABAergic terminals and enhance GABA release.J Neurosci. 2005 Feb 23;25(8):2024-31. doi: 10.1523/JNEUROSCI.4980-04.2005. J Neurosci. 2005. PMID: 15728842 Free PMC article.
-
Presynaptic ionotropic glutamate receptors modulate GABA release in the mouse dorsal motor nucleus of the vagus.Neuroscience. 2015 Nov 12;308:95-105. doi: 10.1016/j.neuroscience.2015.09.009. Epub 2015 Sep 5. Neuroscience. 2015. PMID: 26343294 Free PMC article.
-
Kainate receptors and synaptic plasticity.Neuropharmacology. 2021 Sep 15;196:108540. doi: 10.1016/j.neuropharm.2021.108540. Epub 2021 Mar 30. Neuropharmacology. 2021. PMID: 33794245 Review.
-
Diverse roles for ionotropic glutamate receptors on inhibitory interneurons in developing and adult brain.J Physiol. 2016 Oct 1;594(19):5471-90. doi: 10.1113/JP271764. Epub 2016 May 12. J Physiol. 2016. PMID: 26918438 Free PMC article. Review.
Cited by
-
Perampanel reduces seizure frequency in patients with developmental and epileptic encephalopathy for a long term.Sci Rep. 2024 Dec 3;14(1):30051. doi: 10.1038/s41598-024-82014-5. Sci Rep. 2024. PMID: 39627316 Free PMC article.
-
Calcium-permeable presynaptic AMPA receptors in cerebellar molecular layer interneurones.J Physiol. 2008 Nov 1;586(21):5129-45. doi: 10.1113/jphysiol.2008.159921. Epub 2008 Sep 4. J Physiol. 2008. PMID: 18772200 Free PMC article.
-
Presynaptic AMPA Receptors in Health and Disease.Cells. 2021 Aug 31;10(9):2260. doi: 10.3390/cells10092260. Cells. 2021. PMID: 34571906 Free PMC article. Review.
-
Structure, Function, and Pharmacology of Glutamate Receptor Ion Channels.Pharmacol Rev. 2021 Oct;73(4):298-487. doi: 10.1124/pharmrev.120.000131. Pharmacol Rev. 2021. PMID: 34753794 Free PMC article. Review.
References
-
- Bettler B, Egebjerg J, Sharma G, Pecht G, Hermans-Borgmeyer I, Moll C, Stevens CF, Heinemann S. Cloning of a putative glutamate receptor: a low affinity kainate-binding subunit. Neuron. 1992;8:257–265. - PubMed
-
- Damgaard I, Trenkner E, Sturman JA, Schousboe A. Effect of K+- and kainate-mediated depolarization on survival and functional maturation of GABAergic and glutamatergic neurons in cultures of dissociated mouse cerebellum. Neurochemical Research. 1996;21:267–275. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

