Aggregation of TMV CP plays a role in CP functions and in coat-protein-mediated resistance
- PMID: 17493658
- PMCID: PMC2034504
- DOI: 10.1016/j.virol.2007.03.014
Aggregation of TMV CP plays a role in CP functions and in coat-protein-mediated resistance
Abstract
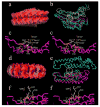
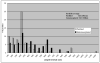
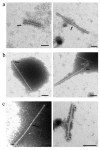
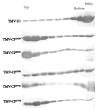
Tobacco mosaic virus (TMV) coat protein (CP) in absence of RNA self-assembles into several different structures depending on pH and ionic strength. Transgenic plants that produce self-assembling CP are resistant to TMV infection, a phenomenon referred to as coat-protein-mediated resistance (CP-MR). The mutant CP Thr42Trp (CP(T42W)) produces enhanced CP-MR compared to wild-type CP. To establish the relationship between the formation of 20S CP aggregates and CP-MR, virus-like particles (VLPs) produced by TMV variants that yield high levels of CP-MR were characterized. We demonstrate that non-helical structures are found in VLPs formed in vivo by CP(T42W) but not by wild-type CP and suggest that the mutation shifts the intracellular equilibrium of aggregates from low to higher proportions of non-helical 20S aggregates. A similar shift in equilibrium of aggregates was observed with CP(D77R), another mutant that confers high level of CP-MR. The mutant CP(D50R) confers a level of CP-MR similar to wild-type CP and aggregates in a manner similar to wild-type CP. We conclude that increased CP-MR is correlated with a shift in intracellular equilibrium of CP aggregates, including aggregates that interfere with virus replication.
Figures

References
-
- Asselin A, Zaitlin M. Characterization of a second protein associated with virions of tobacco mosaic virus. Virology. 1978;91(1):173–81. - PubMed
-
- Bazzini AA, Asurmendi S, Hopp HE, Beachy RN. Tobacco mosaic virus (TMV) and potato virus X (PVX) coat proteins confer heterologous interference to PVX and TMV infection, respectively. J Gen Virol. 2006;87(Pt 4):1005–12. - PubMed
-
- Beachy RN, Zaitlin M. Characterization and in vitro translation of the RNAs from less-than-full-length, virus-related, nucleoprotein rods present in tobacco mosaic virus preparations. Virology. 1977;81(1):160–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

