Transcriptional regulation of the virR operon of the intracellular pathogen Rhodococcus equi
- PMID: 17496078
- PMCID: PMC1951877
- DOI: 10.1128/JB.00431-07
Transcriptional regulation of the virR operon of the intracellular pathogen Rhodococcus equi
Abstract
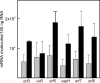
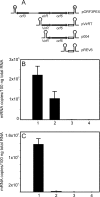
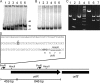
The virR operon, located on the virulence plasmid of the intracellular pathogen Rhodococcus equi, contains five genes, two of which (virR and orf8) encode transcriptional regulators. The first gene of the operon (virR), encoding a LysR-type transcriptional regulator, is transcribed at a constitutive low level, whereas the four downstream genes are induced by low pH and high growth temperature. Differential regulation of the virR operon genes could not be explained by differential mRNA stability, as there were no major differences in mRNA half-lives of the transcripts representing each of the five genes within the virR operon. Transcription of virR is driven by the P(virR) promoter, with a transcription start site 53 bp upstream of the virR initiation codon. The four genes downstream of virR are transcribed from P(virR) and from a second promoter, P(orf5), located 585 bp downstream of the virR initiation codon. VirR binds to a site overlapping the initiation codon of virR, resulting in negative autoregulation of the virR gene, explaining its low constitutive transcription level. The P(orf5) promoter is induced by high temperature and low pH, thus explaining the observed differential gene expression of the virR operon. VirR has a positive effect on P(orf5) activity, whereas the response regulator encoded by orf8 is not involved in regulating transcription of the virR operon. The P(virR) promoter is strikingly similar to those recognized by the principal sigma factors of Streptomyces and Mycobacterium, whereas the P(orf5) promoter does not share sequence similarity with P(virR). This suggests that P(orf5) is recognized by an alternative sigma factor.
Figures


References
-
- Bashyam, M. D., and S. E. Hasnain. 2004. The extracytoplasmic function sigma factors: role in bacterial pathogenesis. Infect. Genet. Evol. 4:301-308. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

