Microvascular network remodeling in dura mater of ovariectomized pigs: role for angiopoietin-1 in estrogen-dependent control of vascular stability
- PMID: 17496211
- PMCID: PMC3332330
- DOI: 10.1152/ajpheart.01156.2006
Microvascular network remodeling in dura mater of ovariectomized pigs: role for angiopoietin-1 in estrogen-dependent control of vascular stability
Abstract
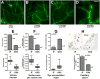
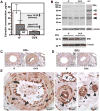
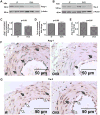
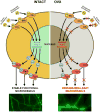
Estrogen is a key regulator of vascular responses and angioadaptation in multiple organs and tissues, including brain. However, the consequences of a loss of ovarian steroid hormone secretion on the status of microvascular networks in brain and meninges are largely unknown. Here, using the perfused dura mater model coupled with high-resolution digital epifluorescence and laser scanning confocal microscopy and computer-assisted morphometric analysis, we demonstrate that cessation of ovarian hormone production causes dramatic vascular remodeling in meningeal microvascular networks characterized by a threefold decrease in microvessel density and capillary rarefaction and an almost fourfold increase in vascular permeability. These changes were accompanied by a significant decrease in angiopoietin-1 (Ang-1) expression and Ang-1/Tie-2 ratio (1.4-fold, P < 0.01, and 1.5-fold, P < 0.05, respectively) in ovariectomized animals compared with intact females, but no changes were detected in the expression of estrogen receptors (ER)-alpha and -beta. We conclude that estrogen-dependent control of Ang-1 expression plays an important role in stabilizing meningeal microvessel and maintaining healthy microvascular networks.
Figures
Similar articles
-
PDGF/VEGF system activation and angiogenesis following initial post ovariectomy meningeal microvessel loss.Cell Cycle. 2008 May 15;7(10):1385-90. doi: 10.4161/cc.7.10.5819. Epub 2008 Feb 25. Cell Cycle. 2008. PMID: 18418080 Free PMC article.
-
Pulsed estrogen therapy prevents post-OVX porcine dura mater microvascular network weakening via a PDGF-BB-dependent mechanism.PLoS One. 2013 Dec 9;8(12):e82900. doi: 10.1371/journal.pone.0082900. eCollection 2013. PLoS One. 2013. PMID: 24349391 Free PMC article.
-
Disruption of Ang-1/Tie-2 signaling contributes to the impaired myocardial vascular maturation and angiogenesis in type II diabetic mice.Arterioscler Thromb Vasc Biol. 2008 Sep;28(9):1606-13. doi: 10.1161/ATVBAHA.108.169235. Epub 2008 Jun 12. Arterioscler Thromb Vasc Biol. 2008. PMID: 18556567 Free PMC article.
-
Vascular integrity mediated by vascular endothelial cadherin and regulated by sphingosine 1-phosphate and angiopoietin-1.Circ J. 2009 Dec;73(12):2183-91. doi: 10.1253/circj.cj-09-0666. Epub 2009 Oct 19. Circ J. 2009. PMID: 19838001 Review.
-
Angiopoietin-1/Tie2 receptor signaling in vascular quiescence and angiogenesis.Histol Histopathol. 2010 Mar;25(3):387-96. doi: 10.14670/HH-25.387. Histol Histopathol. 2010. PMID: 20054809 Review.
Cited by
-
Multi-focus image fusion using epifluorescence microscopy for robust vascular segmentation.Annu Int Conf IEEE Eng Med Biol Soc. 2014;2014:4735-8. doi: 10.1109/EMBC.2014.6944682. Annu Int Conf IEEE Eng Med Biol Soc. 2014. PMID: 25571050 Free PMC article.
-
Gender aspects of CGRP in migraine.Cephalalgia. 2019 Mar;39(3):435-444. doi: 10.1177/0333102417739584. Epub 2017 Oct 30. Cephalalgia. 2019. PMID: 29082826 Free PMC article. Review.
-
A Case Report of Hyperostosis Frontalis Interna.Cureus. 2022 May 13;14(5):e24967. doi: 10.7759/cureus.24967. eCollection 2022 May. Cureus. 2022. PMID: 35698675 Free PMC article.
-
Understanding Streptococcus suis serotype 2 infection in pigs through a transcriptional approach.BMC Genomics. 2011 May 20;12:253. doi: 10.1186/1471-2164-12-253. BMC Genomics. 2011. PMID: 21599948 Free PMC article.
-
Endothelial dysfunction occurs independently of adipose tissue inflammation and insulin resistance in ovariectomized Yucatan miniature-swine.Adipocyte. 2018 Jan 2;7(1):35-44. doi: 10.1080/21623945.2017.1405191. Epub 2017 Dec 28. Adipocyte. 2018. PMID: 29283284 Free PMC article.
References
-
- Albrecht ED, Pepe GJ. Steroid hormone regulation of angiogenesis in the primate endometrium. Front Biosci. 2003;8:d416–d429. - PubMed
-
- Albrecht ED, Robb VA, Pepe GJ. Regulation of placental vascular endothelial growth/permeability factor expression and angiogenesis by estrogen during early baboon pregnancy. J Clin Endocrinol Metab. 2004;89:5803–5809. - PubMed
-
- Ardelt AA, McCullough LD, Korach KS, Wang MM, Munzenmaier DH, Hurn PD. Estradiol regulates angiopoietin-1 mRNA expression through estrogen receptor-α in a rodent experimental stroke model. Stroke. 2005;36:337–341. - PubMed
-
- Baffert F, Le T, Thurston G, McDonald DM. Angiopoietin-1 decreases plasma leakage by reducing number and size of endothelial gaps in venules. Am J Physiol Heart Circ Physiol. 2006;290:H107–H118. - PubMed
-
- Bake S, Sohrabji F. 17beta-estradiol differentially regulates blood-brain barrier permeability in young and aging female rats. Endocrinology. 2004;145:5471–5475. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

