A stirred microchamber for oxygen consumption rate measurements with pancreatic islets
- PMID: 17497731
- PMCID: PMC2859188
- DOI: 10.1002/bit.21486
A stirred microchamber for oxygen consumption rate measurements with pancreatic islets
Abstract
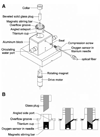
Improvements in pancreatic islet transplantation for treatment of diabetes are hindered by the absence of meaningful islet quality assessment methods. Oxygen consumption rate (OCR) has previously been used to assess the quality of organs and primary tissue for transplantation. In this study, we describe and characterize a stirred microchamber for measuring OCR with small quantities of islets. The device has a titanium body with a chamber volume of about 200 microL and is magnetically stirred and water jacketed for temperature control. Oxygen partial pressure (pO(2)) is measured by fluorescence quenching with a fiber optic probe, and OCR is determined from the linear decrease of pO(2) with time. We demonstrate that measurements can be made rapidly and with high precision. Measurements with betaTC3 cells and islets show that OCR is directly proportional to the number of viable cells in mixtures of live and dead cells and correlate linearly with membrane integrity measurements made with cells that have been cultured for 24 h under various stressful conditions.
Figures





Similar articles
-
Assessment of tissue-engineered islet graft viability by fluorine magnetic resonance spectroscopy.Transplant Proc. 2011 Nov;43(9):3221-5. doi: 10.1016/j.transproceed.2011.09.009. Transplant Proc. 2011. PMID: 22099762 Free PMC article.
-
The use of the BD oxygen biosensor system to assess isolated human islets of langerhans: oxygen consumption as a potential measure of islet potency.Cell Transplant. 2006;15(8-9):745-58. doi: 10.3727/000000006783981440. Cell Transplant. 2006. PMID: 17269445
-
Microchip-based engineering of super-pancreatic islets supported by adipose-derived stem cells.Biomaterials. 2014 Jun;35(17):4815-26. doi: 10.1016/j.biomaterials.2014.02.045. Epub 2014 Mar 15. Biomaterials. 2014. PMID: 24636217
-
The Eye as a Transplantation Site to Monitor Pancreatic Islet Cell Plasticity.Front Endocrinol (Lausanne). 2021 Apr 23;12:652853. doi: 10.3389/fendo.2021.652853. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 33967961 Free PMC article. Review.
-
Effect of oxygen supply on the size of implantable islet-containing encapsulation devices.Panminerva Med. 2016 Mar;58(1):72-7. Epub 2016 Feb 2. Panminerva Med. 2016. PMID: 26837777 Review.
Cited by
-
Antegrade persufflation of porcine kidneys improves renal function after warm ischemia.Front Transplant. 2024 Aug 22;3:1420693. doi: 10.3389/frtra.2024.1420693. eCollection 2024. Front Transplant. 2024. PMID: 39239359 Free PMC article.
-
Mitigating hypoxic stress on pancreatic islets via in situ oxygen generating biomaterial.Biomaterials. 2017 Jun;129:139-151. doi: 10.1016/j.biomaterials.2017.03.018. Epub 2017 Mar 18. Biomaterials. 2017. PMID: 28342320 Free PMC article.
-
Optical sensor arrays designed for guided manufacture of perfluorocarbon nanoemulsions with a non-synthetic stabilizer.Acta Biomater. 2021 Dec;136:558-569. doi: 10.1016/j.actbio.2021.09.038. Epub 2021 Sep 24. Acta Biomater. 2021. PMID: 34563723 Free PMC article.
-
Hypothermic Perfusion Preservation of Pancreas for Islet Grafts: Validation Using a Split Lobe Porcine Model.Cell Med. 2012 Jan 1;2(3):105-110. doi: 10.3727/215517911X617897. Cell Med. 2012. PMID: 24083059 Free PMC article.
-
Long-term viability and function of transplanted islets macroencapsulated at high density are achieved by enhanced oxygen supply.Sci Rep. 2018 Apr 25;8(1):6508. doi: 10.1038/s41598-018-23862-w. Sci Rep. 2018. PMID: 29695723 Free PMC article.
References
-
- Arain S, Svenja W, Heinzle E, Gernot JT, Krause C, Klimant I. Sensing microplates with optodes: Influence of oxygen exchange between sample, air, and plate material. Biotechnol Bioeng. 1998;90:271–280. - PubMed
-
- Avgoustiniatos ES. Ph.D. Thesis. Cambridge: Massachusetts Institute of Technology; 2001. Oxygen diffusion limitation in pancreatic islet culture and immunoisolation.
-
- Bank HL. Assessment of islet cell viability using fluorescent dyes. Diabetologia. 1987;30:812–816. - PubMed
-
- Bank HL. Rapid assessment of islet viability with acridine orange and propidium iodide. In Vitro Cell Dev Biol. 1988;24:266–273. - PubMed
-
- Berridge MV, Tan AS. Characterization of the cellular reduction of 3-(4,5-dimethylthiazol-2- yl)-2,5-diphenyltetrazolium bromide (MTT): Subcellular localization, substrate dependence, and involvement of mitochondrial electron transport in MTT reduction. Arch Biochem Biophys. 1993;303:474–482. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

