Myosin-1a powers the sliding of apical membrane along microvillar actin bundles
- PMID: 17502425
- PMCID: PMC2064212
- DOI: 10.1083/jcb.200701144
Myosin-1a powers the sliding of apical membrane along microvillar actin bundles
Abstract
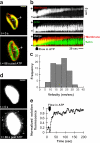
Microvilli are actin-rich membrane protrusions common to a variety of epithelial cell types. Within microvilli of the enterocyte brush border (BB), myosin-1a (Myo1a) forms an ordered ensemble of bridges that link the plasma membrane to the underlying polarized actin bundle. Despite decades of investigation, the function of this unique actomyosin array has remained unclear. Here, we show that addition of ATP to isolated BBs induces a plus end-directed translation of apical membrane along microvillar actin bundles. Upon reaching microvillar tips, membrane is "shed" into solution in the form of small vesicles. Because this movement demonstrates the polarity, velocity, and nucleotide dependence expected for a Myo1a-driven process, and BBs lacking Myo1a fail to undergo membrane translation, we conclude that Myo1a powers this novel form of motility. Thus, in addition to providing a means for amplifying apical surface area, we propose that microvilli function as actomyosin contractile arrays that power the release of BB membrane vesicles into the intestinal lumen.
Figures





Similar articles
-
The enterocyte microvillus is a vesicle-generating organelle.J Cell Biol. 2009 Jun 29;185(7):1285-98. doi: 10.1083/jcb.200902147. J Cell Biol. 2009. PMID: 19564407 Free PMC article.
-
Restoration of cytoskeletal and membrane tethering defects but not defects in membrane trafficking in the intestinal brush border of mice lacking both myosin Ia and myosin VI.Cytoskeleton (Hoboken). 2015 Sep;72(9):455-76. doi: 10.1002/cm.21238. Epub 2015 Sep 16. Cytoskeleton (Hoboken). 2015. PMID: 26286357 Free PMC article.
-
Differential localization and dynamics of class I myosins in the enterocyte microvillus.Mol Biol Cell. 2010 Mar 15;21(6):970-8. doi: 10.1091/mbc.e09-07-0638. Epub 2010 Jan 20. Mol Biol Cell. 2010. PMID: 20089841 Free PMC article.
-
Shaping the intestinal brush border.J Cell Biol. 2014 Nov 24;207(4):441-51. doi: 10.1083/jcb.201407015. J Cell Biol. 2014. PMID: 25422372 Free PMC article. Review.
-
Myosins and membrane trafficking in intestinal brush border assembly.Curr Opin Cell Biol. 2022 Aug;77:102117. doi: 10.1016/j.ceb.2022.102117. Epub 2022 Jul 20. Curr Opin Cell Biol. 2022. PMID: 35870341 Review.
Cited by
-
Myosin 1b Regulates Amino Acid Transport by Associating Transporters with the Apical Plasma Membrane of Kidney Cells.PLoS One. 2015 Sep 11;10(9):e0138012. doi: 10.1371/journal.pone.0138012. eCollection 2015. PLoS One. 2015. PMID: 26361046 Free PMC article.
-
Dynamic length regulation of sensory stereocilia.Semin Cell Dev Biol. 2008 Dec;19(6):502-10. doi: 10.1016/j.semcdb.2008.07.006. Epub 2008 Jul 25. Semin Cell Dev Biol. 2008. PMID: 18692583 Free PMC article. Review.
-
Proteomic analysis of the enterocyte brush border.Am J Physiol Gastrointest Liver Physiol. 2011 May;300(5):G914-26. doi: 10.1152/ajpgi.00005.2011. Epub 2011 Feb 17. Am J Physiol Gastrointest Liver Physiol. 2011. PMID: 21330445 Free PMC article.
-
A new role for the architecture of microvillar actin bundles in apical retention of membrane proteins.Mol Biol Cell. 2012 Jan;23(2):324-36. doi: 10.1091/mbc.E11-09-0765. Epub 2011 Nov 23. Mol Biol Cell. 2012. PMID: 22114352 Free PMC article.
-
Force Generation by Membrane-Associated Myosin-I.Sci Rep. 2016 May 9;6:25524. doi: 10.1038/srep25524. Sci Rep. 2016. PMID: 27156719 Free PMC article.
References
-
- Beaun, A.R., and G. Grondin. 1991. Shedding of vesicular material from the cell surface of eukaryotic cells: different cellular phenomena. Biochim. Biophys. Acta. 1071:203–219. - PubMed
-
- Berg, J.S., and R.E. Cheney. 2002. Myosin-X is an unconventional myosin that undergoes intrafilopodial motility. Nat. Cell Biol. 4:246–250. - PubMed
-
- Black, B.L., Y. Yoneyama, and F. Moog. 1980. Microvillous membrane vesicle accumulation in media during culture of intestine of chick embryo. Biochim. Biophys. Acta. 601:343–348. - PubMed
-
- Bretscher, A. 1991. Microfilament structure and function in the cortical cytoskeleton. Annu. Rev. Cell Biol. 7:337–374. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

