Electrophysiological and immunofluorescence characterization of Ca(2+) channels of acutely isolated rat sphenopalatine ganglion neurons
- PMID: 17507162
- PMCID: PMC1945817
- DOI: 10.1016/j.neulet.2007.04.050
Electrophysiological and immunofluorescence characterization of Ca(2+) channels of acutely isolated rat sphenopalatine ganglion neurons
Abstract
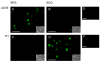
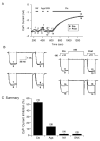
The sphenopalatine ganglion (SPG) is the main parasympathetic ganglion that is involved in regulating cerebral vascular tone and gland secretion. SPG neurons have been implicated in some types of migraine headaches but their precise role has yet to be determined. In addition, very little information is available regarding ion channel modulation by neurotransmitters that are involved in the parasympathetic drive of SPG neurons. In this study, acute isolation of adult rat SPG neurons was developed in order to begin the electrophysiological characterization of this ganglion. Under our dissociation conditions, the average number of neurons obtained per ganglion was greater than 1200. Immunofluorescence imaging results showed positive labeling with acetylcholinesterase (AChE), confirming the parasympathetic nature of SPG neurons. On the other hand, weak tyrosine hydroxylase immunostaining was observed in these neurons. Whole-cell patch-clamp recordings revealed that most of the Ca(2+) current is carried by N-type (53%) and SNX-482 resistant R-type (30%) Ca(2+) channels. In addition, Ca(2+) currents were inhibited in a voltage-dependent manner following exposure to oxotremorine-M (Oxo-M), norepinephrine and ATP via muscarinic acetylcholine receptor 2 (M(2) AChR) subtype, adrenergic and P2Y purinergic receptors, respectively. The peptides VIP and angiotensin II failed to modulate Ca(2+) currents, suggesting that these receptors are not present on the SPG soma or do not couple to Ca(2+) channels. In summary, our data suggest that the Ca(2+) current inhibition mediated by Oxo-M, NE and ATP in adult rat SPG neurons plays an integral part in maintaining parasympathetic control of cranial functions.
Figures

Similar articles
-
Muscarinic acetylcholine receptor modulation of mu (mu) opioid receptors in adult rat sphenopalatine ganglion neurons.J Neurophysiol. 2010 Jan;103(1):172-82. doi: 10.1152/jn.00295.2009. Epub 2009 Nov 4. J Neurophysiol. 2010. PMID: 19889856 Free PMC article.
-
Presynaptic muscarinic M(2)-receptor-mediated inhibition of N-type Ca(2+) channels in cultured sphenopalatine ganglion: direct evidence for acetylcholine inhibition of cerebral nitrergic neurogenic vasodilation.J Pharmacol Exp Ther. 2002 Jul;302(1):397-405. doi: 10.1124/jpet.302.1.397. J Pharmacol Exp Ther. 2002. PMID: 12065743
-
Muscarinic receptor-mediated excitation of rat intracardiac ganglion neurons.Neuropharmacology. 2015 Aug;95:395-404. doi: 10.1016/j.neuropharm.2015.04.014. Epub 2015 Apr 29. Neuropharmacology. 2015. PMID: 25937214
-
N-type Ca2+ channels in cultured rat sphenopalatine ganglion neurons: an immunohistochemical and electrophysiological study.J Cereb Blood Flow Metab. 2000 Jan;20(1):183-91. doi: 10.1097/00004647-200001000-00023. J Cereb Blood Flow Metab. 2000. PMID: 10616807
-
Multiple G-protein-coupled pathways inhibit N-type Ca channels of neurons.Life Sci. 1995;56(11-12):989-92. doi: 10.1016/0024-3205(95)00038-8. Life Sci. 1995. PMID: 10188803 Review.
Cited by
-
Muscarinic acetylcholine receptor modulation of mu (mu) opioid receptors in adult rat sphenopalatine ganglion neurons.J Neurophysiol. 2010 Jan;103(1):172-82. doi: 10.1152/jn.00295.2009. Epub 2009 Nov 4. J Neurophysiol. 2010. PMID: 19889856 Free PMC article.
-
Resection of the nerves bundle from the sphenopalatine ganglia tend to increase the infarction volume following middle cerebral artery occlusion.Neurol Sci. 2010 Aug;31(4):431-5. doi: 10.1007/s10072-010-0238-0. Epub 2010 Mar 3. Neurol Sci. 2010. PMID: 20198495
References
-
- Abbracchio MP, Burnstock G, Boeynaems JM, Barnard EA, Boyer JL, Kennedy C, Knight GE, Fumagalli M, Gachet C, Jacobson KA, Weisman GA. International Union of Pharmacology LVIII: update on the P2Y G protein-coupled nucleotide receptors: from molecular mechanisms and pathophysiology to therapy. Pharmacological Reviews. 2006;58:281–341. - PMC - PubMed
-
- Caulfield MP. Muscarinic receptors--characterization, coupling and function. Pharmacology & Therapeutics. 1993;58:319–379. - PubMed
-
- Ceccatelli S, Lundberg JM, Zhang X, Aman K, Hokfelt T. Immunohistochemical demonstration of nitric oxide synthase in the peripheral autonomic nervous system. Brain Research. 1994;656:381–395. - PubMed
-
- Costa WS, Morais R, Mandarim-De-Lacerda CA. Stereology of the pterygopalatine ganglion of the rat. Italian Journal of Anatomy & Embryology. 1992;97:37–44. - PubMed
-
- Cuevas J, Adams DJ. M4 muscarinic receptor activation modulates calcium channel currents in rat intracardiac neurons. Journal of Neurophysiology. 1997;78:1903–1912. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

