Spc24 and Stu2 promote spindle integrity when DNA replication is stalled
- PMID: 17507656
- PMCID: PMC1949354
- DOI: 10.1091/mbc.e06-09-0882
Spc24 and Stu2 promote spindle integrity when DNA replication is stalled
Abstract
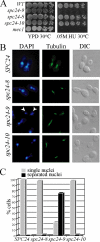
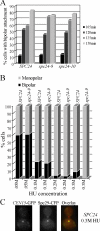
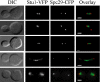
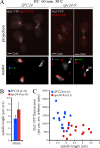
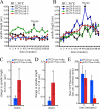
The kinetochore, a protein complex that links chromosomes to microtubules (MTs), is required to prevent spindle expansion during S phase in budding yeast, but the mechanism of how the kinetochore maintains integrity of the bipolar spindle before mitosis is not well understood. Here, we demonstrate that a mutation of Spc24, a component of the conserved Ndc80 kinetochore complex, causes lethality when cells are exposed to the DNA replication inhibitor hydroxyurea (HU) due to premature spindle expansion and segregation of incompletely replicated DNA. Overexpression of Stu1, a CLASP-related MT-associated protein or a truncated form of the XMAP215 orthologue Stu2 rescues spc24-9 HU lethality and prevents spindle expansion. Truncated Stu2 likely acts in a dominant-negative manner, because overexpression of full-length STU2 does not rescue spc24-9 HU lethality, and spindle expansion in spc24-9 HU-treated cells requires active Stu2. Stu1 and Stu2 localize to the kinetochore early in the cell cycle and Stu2 kinetochore localization depends on Spc24. We propose that mislocalization of Stu2 results in premature spindle expansion in S phase stalled spc24-9 mutants. Identifying factors that restrain spindle expansion upon inhibition of DNA replication is likely applicable to the mechanism by which spindle elongation is regulated during a normal cell cycle.
Figures


Similar articles
-
A coordinated kinase and phosphatase network regulates Stu2 recruitment to yeast kinetochores.J Cell Biol. 2025 Aug 4;224(8):e202410196. doi: 10.1083/jcb.202410196. Epub 2025 Jun 27. J Cell Biol. 2025. PMID: 40576560 Free PMC article.
-
Stu1 inversely regulates kinetochore capture and spindle stability.Genes Dev. 2009 Dec 1;23(23):2778-91. doi: 10.1101/gad.541309. Genes Dev. 2009. PMID: 19952112 Free PMC article.
-
Stu2 acts as a microtubule destabilizer in metaphase budding yeast spindles.Mol Biol Cell. 2018 Feb 1;29(3):247-255. doi: 10.1091/mbc.E17-08-0494. Epub 2017 Nov 29. Mol Biol Cell. 2018. PMID: 29187578 Free PMC article.
-
Factors that Control Mitotic Spindle Dynamics.Adv Exp Med Biol. 2017;925:89-101. doi: 10.1007/5584_2016_74. Adv Exp Med Biol. 2017. PMID: 27722958 Review.
-
Kinetochore function: the complications of becoming attached.Curr Biol. 2001 Oct 30;11(21):R855-7. doi: 10.1016/s0960-9822(01)00515-2. Curr Biol. 2001. PMID: 11696340 Review.
Cited by
-
A TOG Protein Confers Tension Sensitivity to Kinetochore-Microtubule Attachments.Cell. 2016 Jun 2;165(6):1428-1439. doi: 10.1016/j.cell.2016.04.030. Epub 2016 May 5. Cell. 2016. PMID: 27156448 Free PMC article.
-
Coupling DNA Replication and Spindle Function in Saccharomyces cerevisiae.Cells. 2021 Nov 30;10(12):3359. doi: 10.3390/cells10123359. Cells. 2021. PMID: 34943867 Free PMC article. Review.
-
A Transcriptomic Insight into the Impact of Colon Cancer Cells on Mast Cells.Int J Mol Sci. 2019 Apr 4;20(7):1689. doi: 10.3390/ijms20071689. Int J Mol Sci. 2019. PMID: 30987352 Free PMC article.
-
A coordinated kinase and phosphatase network regulates Stu2 recruitment to yeast kinetochores.J Cell Biol. 2025 Aug 4;224(8):e202410196. doi: 10.1083/jcb.202410196. Epub 2025 Jun 27. J Cell Biol. 2025. PMID: 40576560 Free PMC article.
-
Hsp110 is required for spindle length control.J Cell Biol. 2012 Aug 20;198(4):623-36. doi: 10.1083/jcb.201111105. J Cell Biol. 2012. PMID: 22908312 Free PMC article.
References
-
- Al-Bassam J., Larsen N. A., Hyman A. A., Harrison S. C. Crystal structure of a TOG domain: conserved features of XMAP215/Dis1-family TOG domains and implications for tubulin binding. Structure. 2007;15:355–362. - PubMed
-
- Allen J. B., Zhou Z., Siede W., Friedberg E. C., Elledge S. J. The SAD1/RAD53 protein kinase controls multiple checkpoints and DNA damage-induced transcription in yeast. Genes Dev. 1994;8:2401–2415. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

