E-cadherin inhibits cell surface localization of the pro-migratory 5T4 oncofetal antigen in mouse embryonic stem cells
- PMID: 17507657
- PMCID: PMC1949355
- DOI: 10.1091/mbc.e06-09-0875
E-cadherin inhibits cell surface localization of the pro-migratory 5T4 oncofetal antigen in mouse embryonic stem cells
Abstract
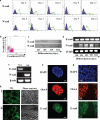
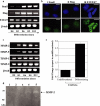
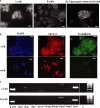
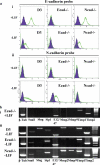
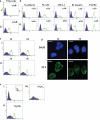
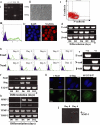
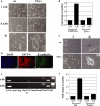
Epithelial-mesenchymal transition (EMT) events occur during embryonic development and are important for the metastatic spread of epithelial tumors. We show here that spontaneous differentiation of mouse embryonic stem (ES) cells is associated with an E- to N-cadherin switch, up-regulation of E-cadherin repressor molecules (Snail and Slug proteins), gelatinase activity (matrix metalloproteinase [MMP]-2 and -9), and increased cellular motility, all characteristic EMT events. The 5T4 oncofetal antigen, previously shown to be associated with very early ES cell differentiation and altered motility, is also a part of this coordinated process. E- and N-cadherin and 5T4 proteins are independently regulated during ES cell differentiation and are not required for induction of EMT-associated transcripts and proteins, as judged from the study of the respective knockout ES cells. Further, abrogation of E-cadherin-mediated cell-cell contact in undifferentiated ES cells using neutralizing antibody results in a reversible mesenchymal phenotype and actin cytoskeleton rearrangement that is concomitant with translocation of the 5T4 antigen from the cytoplasm to the cell surface in an energy-dependent manner. E-cadherin null ES cells are constitutively cell surface 5T4 positive, and although forced expression of E-cadherin cDNA in these cells is sufficient to restore cell-cell contact, cell surface expression of 5T4 antigen is unchanged. 5T4 and N-cadherin knockout ES cells exhibit significantly decreased motility during EMT, demonstrating a functional role for these proteins in this process. We conclude that E-cadherin protein stabilizes cortical actin cytoskeletal arrangement in ES cells, and this can prevent cell surface localization of the promigratory 5T4 antigen.
Figures

Similar articles
-
Epithelial-mesenchymal transition events during human embryonic stem cell differentiation.Cancer Res. 2007 Dec 1;67(23):11254-62. doi: 10.1158/0008-5472.CAN-07-2253. Cancer Res. 2007. PMID: 18056451
-
CXCR4 mediated chemotaxis is regulated by 5T4 oncofetal glycoprotein in mouse embryonic cells.PLoS One. 2010 Apr 1;5(4):e9982. doi: 10.1371/journal.pone.0009982. PLoS One. 2010. PMID: 20376365 Free PMC article.
-
Abrogation of E-cadherin-mediated cell-cell contact in mouse embryonic stem cells results in reversible LIF-independent self-renewal.Stem Cells. 2009 Sep;27(9):2069-80. doi: 10.1002/stem.134. Stem Cells. 2009. PMID: 19544408
-
5T4 oncofoetal antigen: an attractive target for immune intervention in cancer.Cancer Immunol Immunother. 2017 Apr;66(4):415-426. doi: 10.1007/s00262-016-1917-3. Epub 2016 Oct 18. Cancer Immunol Immunother. 2017. PMID: 27757559 Free PMC article. Review.
-
Role of glycogen synthase kinase-3 in cell fate and epithelial-mesenchymal transitions.Cells Tissues Organs. 2007;185(1-3):73-84. doi: 10.1159/000101306. Cells Tissues Organs. 2007. PMID: 17587811 Review.
Cited by
-
Oct-3/4 regulates stem cell identity and cell fate decisions by modulating Wnt/β-catenin signalling.EMBO J. 2010 Oct 6;29(19):3236-48. doi: 10.1038/emboj.2010.200. Epub 2010 Aug 24. EMBO J. 2010. PMID: 20736927 Free PMC article.
-
Inhibition of tumor growth and metastasis by a self-therapeutic nanoparticle.Proc Natl Acad Sci U S A. 2013 Apr 23;110(17):6700-5. doi: 10.1073/pnas.1214547110. Epub 2013 Apr 8. Proc Natl Acad Sci U S A. 2013. PMID: 23569259 Free PMC article.
-
Combination Treatment with an Antibody-Drug Conjugate (A1mcMMAF) Targeting the Oncofetal Glycoprotein 5T4 and Carboplatin Improves Survival in a Xenograft Model of Ovarian Cancer.Target Oncol. 2019 Aug;14(4):465-477. doi: 10.1007/s11523-019-00650-8. Target Oncol. 2019. PMID: 31332693 Free PMC article.
-
Kit K641E oncogene up-regulates Sprouty homolog 4 and trophoblast glycoprotein in interstitial cells of Cajal in a murine model of gastrointestinal stromal tumours.J Cell Mol Med. 2009 Aug;13(8A):1536-48. doi: 10.1111/j.1582-4934.2009.00768.x. Epub 2009 May 19. J Cell Mol Med. 2009. PMID: 19453770 Free PMC article.
-
Upping the ante: recent advances in direct reprogramming.Mol Ther. 2009 Jun;17(6):947-53. doi: 10.1038/mt.2009.72. Epub 2009 Mar 31. Mol Ther. 2009. PMID: 19337233 Free PMC article. Review.
References
-
- Barrallo-Gimeno A., Nieto M. A. The Snail genes as inducers of cell movement and survival: implications in development and cancer. Development. 2005;132:3151–3161. - PubMed
-
- Barrow K. M., Perez-Campo F., Ward C. M. Use of the cytomegalovirus promoter for transient and stable transgene expression in mouse embryonic stem cells. Methods Mol. Biol. 2006;329:283–294. - PubMed
-
- Barrow K. M., Ward C. M., Rutter J., Ali S., Stern P. L. Embryonic expression of murine 5T4 oncofoetal antigen is associated with morphogenetic events at implantation and in developing epithelia. Dev. Dynam. 2005;233:1535–1545. - PubMed
-
- Bates R. C., Mercurio A. M. The epithelial-mesenchymal transition (EMT) and colorectal cancer progression. Cancer Biol. Ther. 2005;4:365–370. - PubMed
-
- Behr R., Heneweer C., Viebahn C., Denker H. W., Thie M. Epithelial-mesenchymal transition in colonies of rhesus monkey embryonic stem cells: a model for processes involved in gastrulation. Stem Cells. 2005;23:805–816. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

