Elimination of antigen-presenting cells and autoreactive T cells by Fas contributes to prevention of autoimmunity
- PMID: 17509906
- PMCID: PMC2575811
- DOI: 10.1016/j.immuni.2007.03.016
Elimination of antigen-presenting cells and autoreactive T cells by Fas contributes to prevention of autoimmunity
Abstract
Fas (also known as Apo-1 and CD95) receptor has been suggested to control T cell expansion by triggering T cell-autonomous apoptosis. This paradigm is based on the extensive lymphoproliferation and systemic autoimmunity in mice and humans lacking Fas or its ligand. However, with systemic loss of Fas, it is unclear whether T cell-extrinsic mechanisms contribute to autoimmunity. We found that tissue-specific deletion of Fas in mouse antigen-presenting cells (APCs) was sufficient to cause systemic autoimmunity, implying that normally APCs are destroyed during immune responses via a Fas-mediated mechanism. Fas expression by APCs was increased by exposure to microbial stimuli. Analysis of mice with Fas loss restricted to T cells revealed that Fas indeed controls autoimmune T cells, but not T cells responding to strong antigenic stimulation. Thus, Fas-dependent elimination of APCs is a major regulatory mechanism curbing autoimmune responses and acts in concert with Fas-mediated regulation of chronically activated autoimmune T cells.
Figures



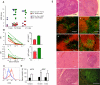
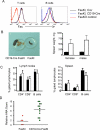

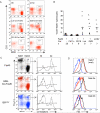
Comment in
-
A FAScinating receptor in self-tolerance.Immunity. 2007 May;26(5):545-7. doi: 10.1016/j.immuni.2007.05.004. Immunity. 2007. PMID: 17521581
References
-
- Arai H, Chan SY, Bishop DK, Nabel GJ. Inhibition of the alloantibody response by CD95 ligand. Nat Med. 1997;3:843–848. - PubMed
-
- Bevan MJ, Fink PJ. The CD8 response on autopilot. Nat Immunol. 2001;2:381–382. - PubMed
-
- Brunner T, Mogil RJ, LaFace D, Yoo NJ, Mahboubi A, Echeverri F, Martin SJ, Force WR, Lynch DH, Ware CF, et al. Cell-autonomous Fas (CD95)/Fas-ligand interaction mediates activation-induced apoptosis in T-cell hybridomas. Nature. 1995;373:441–444. - PubMed
-
- Chen M, Wang YH, Wang Y, Huang L, Sandoval H, Liu YJ, Wang J. Dendritic cell apoptosis in the maintenance of immune tolerance. Science. 2006;311:1160–1164. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

