Live-cell imaging reveals sequential oligomerization and local plasma membrane targeting of stromal interaction molecule 1 after Ca2+ store depletion
- PMID: 17517596
- PMCID: PMC1890489
- DOI: 10.1073/pnas.0702866104
Live-cell imaging reveals sequential oligomerization and local plasma membrane targeting of stromal interaction molecule 1 after Ca2+ store depletion
Abstract
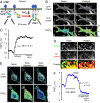
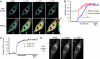
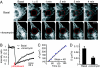
Stromal interaction molecule 1 (STIM1) has recently been identified by our group and others as an endoplasmic reticulum (ER) Ca(2+) sensor that responds to ER Ca(2+) store depletion and activates Ca(2+) channels in the plasma membrane (PM). The molecular mechanism by which STIM1 transduces signals from the ER lumen to the PM is not yet understood. Here we developed a live-cell FRET approach and show that STIM1 forms oligomers within 5 s after Ca(2+) store depletion. These oligomers rapidly dissociated when ER Ca(2+) stores were refilled. We further show that STIM1 formed oligomers before its translocation within the ER network to ER-PM junctions. A mutant STIM1 lacking the C-terminal polybasic PM-targeting motif oligomerized after Ca(2+) store depletion but failed to form puncta at ER-PM junctions. Using fluorescence recovery after photobleaching measurements to monitor STIM1 mobility, we show that STIM1 oligomers translocate on average only 2 mum to reach ER-PM junctions, arguing that STIM1 ER-to-PM signaling is a local process that is suitable for generating cytosolic Ca(2+) gradients. Together, our live-cell measurements dissect the STIM1 ER-to-PM signaling relay into four sequential steps: (i) dissociation of Ca(2+), (ii) rapid oligomerization, (iii) spatially restricted translocation to nearby ER-PM junctions, and (iv) activation of PM Ca(2+) channels.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
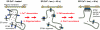
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

