BimS-induced apoptosis requires mitochondrial localization but not interaction with anti-apoptotic Bcl-2 proteins
- PMID: 17517961
- PMCID: PMC2064208
- DOI: 10.1083/jcb.200610148
BimS-induced apoptosis requires mitochondrial localization but not interaction with anti-apoptotic Bcl-2 proteins
Abstract
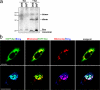
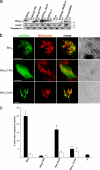
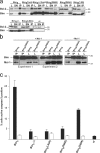
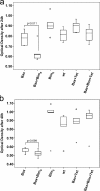
Release of apoptogenic proteins such as cytochrome c from mitochondria is regulated by pro- and anti-apoptotic Bcl-2 family proteins, with pro-apoptotic BH3-only proteins activating Bax and Bak. Current models assume that apoptosis induction occurs via the binding and inactivation of anti-apoptotic Bcl-2 proteins by BH3-only proteins or by direct binding to Bax. Here, we analyze apoptosis induction by the BH3-only protein Bim(S). Regulated expression of Bim(S) in epithelial cells was followed by its rapid mitochondrial translocation and mitochondrial membrane insertion in the absence of detectable binding to anti-apoptotic Bcl-2 proteins. This caused mitochondrial recruitment and activation of Bax and apoptosis. Mutational analysis of Bim(S) showed that mitochondrial targeting, but not binding to Bcl-2 or Mcl-1, was required for apoptosis induction. In yeast, Bim(S) enhanced the killing activity of Bax in the absence of anti-apoptotic Bcl-2 proteins. Thus, cell death induction by a BH3-only protein can occur through a process that is independent of anti-apoptotic Bcl-2 proteins but requires mitochondrial targeting.
Figures



References
-
- Adams, J.M. 2003. Ways of dying: multiple pathways to apoptosis. Genes Dev. 17:2481–2495. - PubMed
-
- Cartron, P.F., H. Arokium, L. Oliver, K. Meflah, S. Manon, and F.M. Vallette. 2005. Distinct domains control the addressing and the insertion of Bax into mitochondria. J. Biol. Chem. 280:10587–10598. - PubMed
-
- Certo, M., G. Moore Vdel, M. Nishino, G. Wei, S. Korsmeyer, S.A. Armstrong, and A. Letai. 2006. Mitochondria primed by death signals determine cellular addiction to antiapoptotic BCL-2 family members. Cancer Cell. 9:351–365. - PubMed
-
- Chen, L., S.N. Willis, A. Wei, B.J. Smith, J.I. Fletcher, M.G. Hinds, P.M. Colman, C.L. Day, J.M. Adams, and D.C. Huang. 2005. Differential targeting of prosurvival Bcl-2 proteins by their BH3-only ligands allows complementary apoptotic function. Mol. Cell. 17:393–403. - PubMed
-
- Cory, S., and J.M. Adams. 2002. The Bcl2 family: regulators of the cellular life-or-death switch. Nat. Rev. Cancer. 2:647–656. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

