Activation of the mTOR pathway in sporadic angiomyolipomas and other perivascular epithelioid cell neoplasms
- PMID: 17521703
- PMCID: PMC2722219
- DOI: 10.1016/j.humpath.2007.01.028
Activation of the mTOR pathway in sporadic angiomyolipomas and other perivascular epithelioid cell neoplasms
Abstract
Angiomyolipoma (AML) belong to a family of tumors known as perivascular epithelioid cell tumors (PEComas) that share a common immunophenotypic profile of muscle and melanocytic differentiation. These tumors are clonal in nature and have a strong association with tuberous sclerosis. Genetic analyses have reported allelic imbalance at the TSC2 locus on 16p13. In the context of non-tuberous sclerosis complex (TSC), non-lymphangioleiomyomatosis-associated AMLs, and non-renal PEComas, the functional status of the TSC2 signaling pathway has not been reported. Studies over the last several years have uncovered a critical role of the TSC1/2 genes in negatively regulating the Rheb/mTOR/p70S6K cascade. Here, we examined the activity of this pathway in sporadic AMLs and PEComas using immunohistochemical and biochemical analyses. We found increased levels of phospho-p70S6K, a marker of mTOR activity, in 15 of 15 non-TSC AMLs. This was accompanied by reduced phospho-AKT expression, a pattern that is consistent with the disruption of TSC1/2 function. Western blot analysis confirmed mTOR activation concurrent with the loss of TSC2 and not TSC1 in sporadic AMLs. Similarly, elevated phospho-p70S6K and reduced phospho-AKT expression was detected in 14 of 15 cases of extrarenal PEComas. These observations provide the first functional evidence that mTOR activation is common to sporadic, non-TSC-related AMLs and PEComas. This suggests the possibility that mTOR inhibitors such as rapamycin may be therapeutic for this class of disease.
Figures


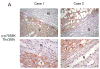




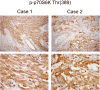
References
-
- Folpe AL. Neoplasms with perivascular epitheloid cell differentiation (PEComas) In: Fletcher CDMUK, Mertens F, editors. World Health Organization Classification of Tumors: Pathology and Genetics of Tumors of Soft Tissue and Bone. Lyon: IARC Press; 2002. pp. 221–222.
-
- Hornick JL, Fletcher CD. PEComa: what do we know so far? Histopathology. 2006;48:75–82. - PubMed
-
- Gomez MR. Definition and criteria for diagnosis. In: Gomez MR, Sampson JR, Whittemore VH, editors. Tuberous Sclerosis Complex. New York: Oxford University Press; 1999. pp. 10–23.
-
- Crino PB, Nathanson KL, Henske EP. The tuberous sclerosis complex. N Engl J Med. 2006;355:1345–1356. - PubMed
-
- Costello LC, Hartman TE, Ryu JH. High frequency of pulmonary lymphangioleiomyomatosis in women with tuberous sclerosis complex. Mayo Clin Proc. 2000;75:591–594. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

