Multiple dose-dependent roles for Sox2 in the patterning and differentiation of anterior foregut endoderm
- PMID: 17522155
- PMCID: PMC3625644
- DOI: 10.1242/dev.003855
Multiple dose-dependent roles for Sox2 in the patterning and differentiation of anterior foregut endoderm
Abstract
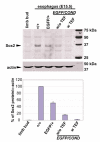
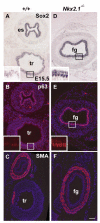
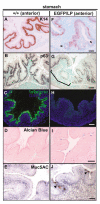
Sox2 is expressed in developing foregut endoderm, with highest levels in the future esophagus and anterior stomach. By contrast, Nkx2.1 (Titf1) is expressed ventrally, in the future trachea. In humans, heterozygosity for SOX2 is associated with anopthalmia-esophageal-genital syndrome (OMIM 600992), a condition including esophageal atresia (EA) and tracheoesophageal fistula (TEF), in which the trachea and esophagus fail to separate. Mouse embryos heterozygous for the null allele, Sox2(EGFP), appear normal. However, further reductions in Sox2, using Sox2(LP) and Sox2(COND) hypomorphic alleles, result in multiple abnormalities. Approximately 60% of Sox2(EGFP/COND) embryos have EA with distal TEF in which Sox2 is undetectable by immunohistochemistry or western blot. The mutant esophagus morphologically resembles the trachea, with ectopic expression of Nkx2.1, a columnar, ciliated epithelium, and very few p63(+) basal cells. By contrast, the abnormal foregut of Nkx2.1-null embryos expresses elevated Sox2 and p63, suggesting reciprocal regulation of Sox2 and Nkx2.1 during early dorsal/ventral foregut patterning. Organ culture experiments further suggest that FGF signaling from the ventral mesenchyme regulates Sox2 expression in the endoderm. In the 40% Sox2(EGFP/COND) embryos in which Sox2 levels are approximately 18% of wild type there is no TEF. However, the esophagus is still abnormal, with luminal mucus-producing cells, fewer p63(+) cells, and ectopic expression of genes normally expressed in glandular stomach and intestine. In all hypomorphic embryos the forestomach has an abnormal phenotype, with reduced keratinization, ectopic mucus cells and columnar epithelium. These findings suggest that Sox2 plays a second role in establishing the boundary between the keratinized, squamous esophagus/forestomach and glandular hindstomach.
Figures





References
-
- Aubin J, Dery U, Lemieux M, Chailler P, Jeannotte L. Stomach regional specification requires Hoxa5-driven mesenchymal-epithelial signaling. Development. 2002;129:4075–4087. - PubMed
-
- Brunner HG, van Bokhoven H. Genetic players in esophageal atresia and tracheoesophageal fistula. Curr. Opin. Genet. Dev. 2005;15:341–347. - PubMed
-
- Calmont A, Wandzioch E, Tremblay KD, Minowada G, Kaestner KH, Martin GR, Zaret KS. An FGF response pathway that mediates hepatic gene induction in embryonic endoderm cells. Dev. Cell. 2006;11:339–348. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

