Protein kinase PKR plays a stimulus- and virus-dependent role in apoptotic death and virus multiplication in human cells
- PMID: 17522227
- PMCID: PMC1951329
- DOI: 10.1128/JVI.00426-07
Protein kinase PKR plays a stimulus- and virus-dependent role in apoptotic death and virus multiplication in human cells
Abstract
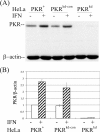
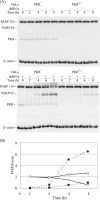
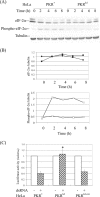
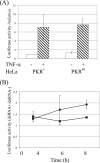
The protein kinase regulated by double-stranded RNA (dsRNA), PKR, is implicated in a range of biologic processes, including apoptotic death and interferon antiviral responses, based in part on studies with mouse cells genetically deficient in Pkr. To test the role of the PKR protein in human cells, an RNA interference silencing strategy was used to generate stable HeLa cell lines with less than 2% of the PKR protein (PKR deficient) compared to either parental or control knockdown HeLa lines. Phosphorylation of the alpha subunit of eukaryotic initiation factor 2 on serine 51 was not detectably increased in response to dsRNA in PKR-deficient HeLa cells but was elevated severalfold in PKR-sufficient cells. PKR-deficient cells displayed reduced dsRNA-induced apoptosis compared to PKR-sufficient cell lines, whereas tumor necrosis factor alpha (TNF-alpha)-induced apoptosis was comparable between the HeLa lines. NF-kappaB was activated to a comparable extent in PKR-deficient and PKR-sufficient HeLa cells upon treatment with either dsRNA or TNF-alpha. The antiviral response against vesicular stomatitis virus was reduced in interferon-treated PKR-deficient compared to PKR-sufficient HeLa cells. However, the growth of two human viruses, adenovirus and reovirus, was unaffected by the PKR knockdown. Surprisingly, the yield of mutant adenovirus that fails to encode VAI RNA was not enhanced in PKR-deficient cells, indicating the importance of host factors in addition to PKR in conferring the VAI RNA phenotype.
Figures



Similar articles
-
Loss of protein kinase PKR expression in human HeLa cells complements the vaccinia virus E3L deletion mutant phenotype by restoration of viral protein synthesis.J Virol. 2008 Jan;82(2):840-8. doi: 10.1128/JVI.01891-07. Epub 2007 Oct 24. J Virol. 2008. PMID: 17959656 Free PMC article.
-
Phosphorylation of eukaryotic translation initiation factor 2 mediates apoptosis in response to activation of the double-stranded RNA-dependent protein kinase.J Biol Chem. 1998 Jan 23;273(4):2416-23. doi: 10.1074/jbc.273.4.2416. J Biol Chem. 1998. PMID: 9442091
-
Interaction between PKR and PACT mediated by LPS-inducible NF-κB in human gingival cells.J Cell Biochem. 2012 Jan;113(1):165-73. doi: 10.1002/jcb.23340. J Cell Biochem. 2012. PMID: 21882225
-
The dsRNA protein kinase PKR: virus and cell control.Biochimie. 2007 Jun-Jul;89(6-7):799-811. doi: 10.1016/j.biochi.2007.03.001. Epub 2007 Mar 12. Biochimie. 2007. PMID: 17451862 Review.
-
PKR--a protein kinase regulated by double-stranded RNA.Int J Biochem Cell Biol. 1997 Jul;29(7):945-9. doi: 10.1016/s1357-2725(96)00169-0. Int J Biochem Cell Biol. 1997. PMID: 9375375 Review.
Cited by
-
Overexpression of eIF5 or its protein mimic 5MP perturbs eIF2 function and induces ATF4 translation through delayed re-initiation.Nucleic Acids Res. 2016 Oct 14;44(18):8704-8713. doi: 10.1093/nar/gkw559. Epub 2016 Jun 20. Nucleic Acids Res. 2016. PMID: 27325740 Free PMC article.
-
Severe acute respiratory syndrome coronavirus triggers apoptosis via protein kinase R but is resistant to its antiviral activity.J Virol. 2009 Mar;83(5):2298-309. doi: 10.1128/JVI.01245-08. Epub 2008 Dec 24. J Virol. 2009. PMID: 19109397 Free PMC article.
-
PKR Transduces MDA5-Dependent Signals for Type I IFN Induction.PLoS Pathog. 2016 Mar 3;12(3):e1005489. doi: 10.1371/journal.ppat.1005489. eCollection 2016 Mar. PLoS Pathog. 2016. PMID: 26939124 Free PMC article.
-
Editing of Cellular Self-RNAs by Adenosine Deaminase ADAR1 Suppresses Innate Immune Stress Responses.J Biol Chem. 2016 Mar 18;291(12):6158-68. doi: 10.1074/jbc.M115.709014. Epub 2016 Jan 27. J Biol Chem. 2016. PMID: 26817845 Free PMC article.
-
RNA adenosine deaminase ADAR1 deficiency leads to increased activation of protein kinase PKR and reduced vesicular stomatitis virus growth following interferon treatment.Virology. 2010 Jan 20;396(2):316-22. doi: 10.1016/j.virol.2009.10.026. Epub 2009 Nov 12. Virology. 2010. PMID: 19913273 Free PMC article.
References
-
- Abraham, N., D. F. Stojdl, P. I. Duncan, N. Methot, T. Ishii, M. Dube, B. C. Vanderhyden, H. L. Atkins, D. A. Gray, M. W. McBurney, A. E. Koromilas, E. G. Brown, N. Sonenberg, and J. C. Bell. 1999. Characterization of transgenic mice with targeted disruption of the catalytic domain of the double-stranded RNA-dependent protein kinase, PKR. J. Biol. Chem. 274:5953-5962. - PubMed
-
- Alexopoulou, L., A. C. Holt, R. Medzhitov, and R. A. Flavell. 2001. Recognition of double-stranded RNA and activation of NF-κB by Toll-like receptor 3. Nature 413:732-738. - PubMed
-
- Ashkenazi, A., and V. M. Dixit. 1998. Death receptors: signaling and modulation. Science 281:1305-1308. - PubMed
-
- Baltzis, D., S. Li, and A. E. Koromilas. 2002. Functional characterization of pkr gene products expressed in cells from mice with a targeted deletion of the N terminus or C terminus domain of PKR. J. Biol. Chem. 277:38364-38372. - PubMed
-
- Barber, G. N. 2005. The dsRNA-dependent protein kinase, PKR and cell death. Cell Death Differ. 12:563-570. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

