Clathrin-dependent entry of severe acute respiratory syndrome coronavirus into target cells expressing ACE2 with the cytoplasmic tail deleted
- PMID: 17522231
- PMCID: PMC1951348
- DOI: 10.1128/JVI.00253-07
Clathrin-dependent entry of severe acute respiratory syndrome coronavirus into target cells expressing ACE2 with the cytoplasmic tail deleted
Abstract
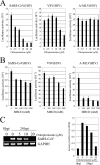
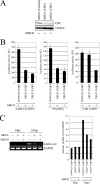
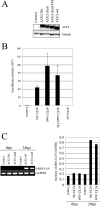
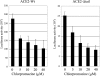
The penetration of various viruses into host cells is accomplished by hijacking the host endocytosis machinery. In the case of severe acute respiratory syndrome coronavirus (SARS-CoV) infection, viral entry is reported to require a low pH in intracytoplasmic vesicles; however, little is known about how SARS-CoV invades such compartments. Here we demonstrate that SARS-CoV mainly utilizes the clathrin-mediated endocytosis pathway for its entry to target cells by using infectious SARS-CoV, as well as a SARS-CoV pseudovirus packaged in the SARS-CoV envelope. The SARS-CoV entered caveolin-1-negative HepG2 cells, and the entry was significantly inhibited by treatment with chlorpromazine, an inhibitor for clathrin-dependent endocytosis, and by small interfering RNA-mediated gene silencing for the clathrin heavy chain. Furthermore, the SARS-CoV entered COS7 cells transfected with the mutant of ACE2 with the cytoplasmic tail deleted, SARS-CoV receptor, as well as the wild-type ACE2, and their entries were significantly inhibited by treatment with chlorpromazine. In addition, ACE2 translocated into EEA1-positive early endosomes immediately after the virus attachment to ACE2. These results suggest that when SARS-CoV binds ACE2 it is internalized and penetrates early endosomes in a clathrin-dependent manner and that the cytoplasmic tail of ACE2 is not required for the penetration of SARS-CoV.
Figures

References
-
- Belleudi, F., V. Visco, M. Ceridono, L. Leone, R. Muraro, L. Frati, and M. R. Torrisi. 2003. Ligand-induced clathrin-mediated endocytosis of the keratinocyte growth factor receptor occurs independently of either phosphorylation or recruitment of eps15. FEBS Lett. 553:262-270. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

