Artemether-lumefantrine versus dihydroartemisinin-piperaquine for treatment of malaria: a randomized trial
- PMID: 17525792
- PMCID: PMC1876597
- DOI: 10.1371/journal.pctr.0020020
Artemether-lumefantrine versus dihydroartemisinin-piperaquine for treatment of malaria: a randomized trial
Abstract
Objectives: To compare the efficacy and safety of artemether-lumefantrine (AL) and dihydroartemisinin-piperaquine (DP) for treating uncomplicated falciparum malaria in Uganda.
Design: Randomized single-blinded clinical trial.
Setting: Apac, Uganda, an area of very high malaria transmission intensity.
Participants: Children aged 6 mo to 10 y with uncomplicated falciparum malaria.
Intervention: Treatment of malaria with AL or DP, each following standard 3-d dosing regimens.
Outcome measures: Risks of recurrent parasitemia at 28 and 42 d, unadjusted and adjusted by genotyping to distinguish recrudescences and new infections.
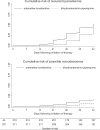
Results: Of 421 enrolled participants, 417 (99%) completed follow-up. The unadjusted risk of recurrent falciparum parasitemia was significantly lower for participants treated with DP than for those treated with AL after 28 d (11% versus 29%; risk difference [RD] 18%, 95% confidence interval [CI] 11%-26%) and 42 d (43% versus 53%; RD 9.6%, 95% CI 0%-19%) of follow-up. Similarly, the risk of recurrent parasitemia due to possible recrudescence (adjusted by genotyping) was significantly lower for participants treated with DP than for those treated with AL after 28 d (1.9% versus 8.9%; RD 7.0%, 95% CI 2.5%-12%) and 42 d (6.9% versus 16%; RD 9.5%, 95% CI 2.8%-16%). Patients treated with DP had a lower risk of recurrent parasitemia due to non-falciparum species, development of gametocytemia, and higher mean increase in hemoglobin compared to patients treated with AL. Both drugs were well tolerated; serious adverse events were uncommon and unrelated to study drugs.
Conclusion: DP was superior to AL for reducing the risk of recurrent parasitemia and gametocytemia, and provided improved hemoglobin recovery. DP thus appears to be a good alternative to AL as first-line treatment of uncomplicated malaria in Uganda. To maximize the benefit of artemisinin-based combination therapy in Africa, treatment should be integrated with aggressive strategies to reduce malaria transmission intensity.
Conflict of interest statement
Figures
References
-
- Attaran A, Barnes KI, Curtis C, d'Alessandro U, Fanello CI, et al. WHO, the Global Fund, and medical malpractice in malaria treatment. Lancet. 2004;363:237–240. - PubMed
-
- Piola P, Fogg C, Bajunirwe F, Biraro S, Grandesso F, et al. Supervised versus unsupervised intake of six-dose artemether-lumefantrine for treatment of acute, uncomplicated Plasmodium falciparum malaria in Mbarara, Uganda: A randomised trial. Lancet. 2005;365:1467–1473. - PubMed
-
- Martensson A, Stromberg J, Sisowath C, Msellem MI, Gil JP, et al. Efficacy of artesunate plus amodiaquine versus that of artemether-lumefantrine for the treatment of uncomplicated childhood Plasmodium falciparum malaria in Zanzibar, Tanzania. Clin Infect Dis. 2005;41:1079–1086. - PubMed
-
- Mutabingwa TK, Anthony D, Heller A, Hallett R, Ahmed J, et al. Amodiaquine alone, amodiaquine+sulfadoxine-pyrimethamine, amodiaquine+artesunate, and artemether-lumefantrine for outpatient treatment of malaria in Tanzanian children: A four-arm randomised effectiveness trial. Lancet. 2005;365:1474–1480. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous