Establishing a definitive stoichiometry for the Na+/monocarboxylate cotransporter SMCT1
- PMID: 17526579
- PMCID: PMC1965447
- DOI: 10.1529/biophysj.107.108555
Establishing a definitive stoichiometry for the Na+/monocarboxylate cotransporter SMCT1
Abstract
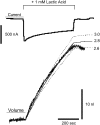
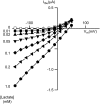
Several different stoichiometries have been proposed for the Na(+)/monocarboxylate cotransporter SMCT1, including variable Na(+)/substrate stoichiometry. In this work, we have definitively established an invariant 2:1 cotransport stoichiometry for SMCT1. By using two independent means of assay, we first showed that SMCT1 exhibits a 2:1 stoichiometry for Na(+)/lactate cotransport. Radiolabel uptake experiments proved that, unlike lactate, propionic acid diffuses passively through oocyte membranes and, consequently, propionate is a poor candidate for stoichiometric determination by these methods. Although we previously determined SMCT1 stoichiometry by measuring reversal potentials, this technique produced erroneous values, because SMCT1 simultaneously mediates both an inwardly rectifying cotransport current and an outwardly rectifying anionic leak current; the leak current predominates in the range where reversal potentials are observed. We therefore employed a method that compared the effect of halving the external Na(+) concentration to the effect of halving the external substrate concentration on zero-current potentials. Both lactate and propionate were cotransported through SMCT1 using 2:1 stoichiometries. The leak current passing through the protein has a 1 osmolyte/charge stoichiometry. Identification of cotransporter stoichiometry is not always a trivial task and it can lead to a much better understanding of the transport activity mediated by the protein in question.
Figures

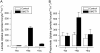
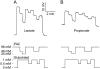
Similar articles
-
The human tumour suppressor gene SLC5A8 expresses a Na+-monocarboxylate cotransporter.J Physiol. 2004 Jun 15;557(Pt 3):719-31. doi: 10.1113/jphysiol.2004.063859. Epub 2004 Apr 16. J Physiol. 2004. PMID: 15090606 Free PMC article.
-
Identity of SMCT1 (SLC5A8) as a neuron-specific Na+-coupled transporter for active uptake of L-lactate and ketone bodies in the brain.J Neurochem. 2006 Jul;98(1):279-88. doi: 10.1111/j.1471-4159.2006.03878.x. J Neurochem. 2006. PMID: 16805814
-
Insulin and SGK1 reduce the function of Na+/monocarboxylate transporter 1 (SMCT1/SLC5A8).Am J Physiol Cell Physiol. 2016 Nov 1;311(5):C720-C734. doi: 10.1152/ajpcell.00104.2015. Epub 2016 Aug 3. Am J Physiol Cell Physiol. 2016. PMID: 27488665 Free PMC article.
-
Anionic leak currents through the Na+/monocarboxylate cotransporter SMCT1.Am J Physiol Cell Physiol. 2010 Jan;298(1):C124-31. doi: 10.1152/ajpcell.00220.2009. Epub 2009 Oct 28. Am J Physiol Cell Physiol. 2010. PMID: 19864324
-
Transport characteristics of 5-aminosalicylic acid into colonic epithelium: Involvement of sodium-coupled monocarboxylate transporter SMCT1-mediated transport system.Biochem Biophys Res Commun. 2020 Apr 9;524(3):561-566. doi: 10.1016/j.bbrc.2020.01.139. Epub 2020 Jan 31. Biochem Biophys Res Commun. 2020. PMID: 32014250
Cited by
-
Distribution of glucose transporters in renal diseases.J Biomed Sci. 2017 Aug 31;24(1):64. doi: 10.1186/s12929-017-0371-7. J Biomed Sci. 2017. PMID: 28854935 Free PMC article. Review.
-
The ABCs of membrane transporters in health and disease (SLC series): introduction.Mol Aspects Med. 2013 Apr-Jun;34(2-3):95-107. doi: 10.1016/j.mam.2012.12.009. Mol Aspects Med. 2013. PMID: 23506860 Free PMC article. Review.
-
Microbiota-microglia connections in age-related cognition decline.Aging Cell. 2022 May;21(5):e13599. doi: 10.1111/acel.13599. Epub 2022 Mar 29. Aging Cell. 2022. PMID: 35349746 Free PMC article. Review.
-
EmrE reminds us to expect the unexpected in membrane transport.J Gen Physiol. 2020 Jan 6;152(1):e201912467. doi: 10.1085/jgp.201912467. J Gen Physiol. 2020. PMID: 31816640 Free PMC article.
-
Nutrient transport in the mammary gland: calcium, trace minerals and water soluble vitamins.J Mammary Gland Biol Neoplasia. 2014 Mar;19(1):73-90. doi: 10.1007/s10911-014-9317-9. Epub 2014 Feb 25. J Mammary Gland Biol Neoplasia. 2014. PMID: 24567109 Review.
References
-
- Turner, R. J., and A. Moran. 1982. Further studies of proximal tubular brush border membrane D-glucose transport heterogeneity. J. Membr. Biol. 70:37–45. - PubMed
-
- Wright, E. M., and E. Turk. 2004. The sodium/glucose cotransport family SLC5. Pflugers Arch. 447:510–518. - PubMed
-
- Chen, N. H., M. E. Reith, and M. W. Quick. 2004. Synaptic uptake and beyond: the sodium- and chloride-dependent neurotransmitter transporter family SLC6. Pflugers Arch. 447:519–531. - PubMed
-
- Coady, M. J., X. Z. Chen, and J. Y. Lapointe. 1996. rBAT is an amino acid exchanger with variable stoichiometry. J. Membr. Biol. 149:1–8. - PubMed
-
- Alonso, G. L., D. A. Gonzalez, D. Takara, M. A. Ostuni, and G. A. Sanchez. 2001. Kinetic analysis of a model of the sarcoplasmic reticulum Ca-ATPase, with variable stoichiometry, which enhances the amount and the rate of Ca transport. J. Theor. Biol. 208:251–260. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

