Functional characterization of genetic variants of human FMO3 associated with trimethylaminuria
- PMID: 17531949
- PMCID: PMC2039921
- DOI: 10.1016/j.abb.2007.04.014
Functional characterization of genetic variants of human FMO3 associated with trimethylaminuria
Abstract
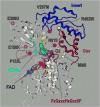
Impaired conversion of trimethylamine to trimethylamine N-oxide by human flavin containing monooxygenase 3 (FMO3) is strongly associated with primary trimethylaminuria, also known as 'fish-odor' syndrome. Numerous non-synonymous mutations in FMO3 have been identified in patients suffering from this metabolic disorder (e.g., N61S, M66I, P153L, and R492W), but the molecular mechanism(s) underlying the functional deficit attributed to these alleles has not been elucidated. The purpose of the present study was to determine the impact of these disease-associated genetic variants on FMO3 holoenzyme formation and on steady-state kinetic parameters for metabolism of several substrates, including trimethylamine. For comparative purposes, several common allelic variants not associated with primary trimethylaminuria (i.e., E158K, V257M, E308G, and the E158K/E308G haplotype) were also analyzed. When recombinantly expressed in insect cells, only the M66I and R492W mutants failed to incorporate/retain the FAD cofactor. Of the remaining mutant proteins P153L and N61S displayed substantially reduced (<10%) catalytic efficiencies for trimethylamine N-oxygenation relative to the wild-type enzyme. For N61S, reduced catalytic efficiency was solely a consequence of an increased K(m), whereas for P153L, both K(m) and k(cat) were altered. Similar results were obtained when benzydamine N-oxygenation was monitored. A homology model for FMO3 was constructed based on the crystal structure for yeast FMO which places the N61 residue alone, of the mutants analyzed here, in close proximity to the FAD catalytic center. These data demonstrate that primary trimethylaminuria is multifactorial in origin in that enzyme dysfunction can result from kinetic incompetencies as well as impaired assembly of holoprotein.
Figures
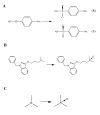


 ), P153L (▲) and N61S (●) catalyzed TMA N-oxygenation.
), P153L (▲) and N61S (●) catalyzed TMA N-oxygenation.
References
-
- Holmes HC, Burns SP, Michelakakis H, Kordoni V, Bain MD, Chalmers RA, Rafter JE, Iles RA. Choline and L-carnitine as precursors of trimethylamine. Biochem Soc Trans. 1997;25:96S. - PubMed
-
- Tjoa S, Fennessey P. The identification of trimethylamine excess in man: quantitative analysis and biochemical origins. Anal Biochem. 1991;197:77–82. - PubMed
-
- Ruocco V, Florio M, Filioli FG, Guerrera V, Prota G. An unusual case of trimethylaminuria. Br J Dermatol. 1989;120:459–461. - PubMed
-
- Lin J, Cashman JR. Detoxication of tyramine by the flavin-containing monooxygenase: stereoselective formation of the trans oxime. Chem Res Toxicol. 1997;10:842–852. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

