Forebrain ischemia-reperfusion simulating cardiac arrest in mice induces edema and DNA fragmentation in the brain
- PMID: 17532882
- PMCID: PMC2644455
Forebrain ischemia-reperfusion simulating cardiac arrest in mice induces edema and DNA fragmentation in the brain
Abstract
Brain injury affects one-third of persons who survive after heart attack, even with restoration of spontaneous circulation by cardiopulmonary resuscitation. We studied brain injury resulting from transient bilateral carotid artery occlusion (BCAO) and reperfusion by simulating heart attack and restoration of circulation, respectively, in live C57Black6 mice. This model is known to induce neuronal death in the hippocampus, striatum, and cortex. We report the appearance of edema after transient BCAO of 60 minutes and 1 day of reperfusion. Hyperintensity in diffusion-weighted magnetic resonance imaging (MRI) was detectable in the striatum, thalamus, and cortex but not in the hippocampus. To determine whether damage to the hippocampus can be detected in live animals, we infused a T(2) susceptibility magnetic resonance contrast agent (superparamagnetic iron oxide nanoparticles [SPIONs]) that was linked to single-stranded deoxyribonucleic acid (DNA) complementary in sequence to c-fos messenger ribonucleic acid (SPION-cfos); we acquired in vivo T(2)*-weighted MRI 3 days later. SPION retention was measured as T(2)* (milliseconds) signal reduction or R(2)* value (s(-1)) elevation. We found that animals treated with 60-minute BCAO and 7-day reperfusion exhibited significantly less SPION retention in the hippocampus and cortex than sham-operated animals. These findings suggest that brain injury induced by cardiac arrest can be detected in live animals.
Figures


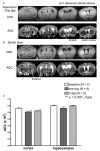
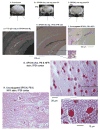
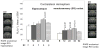

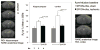
Similar articles
-
MR contrast probes that trace gene transcripts for cerebral ischemia in live animals.FASEB J. 2007 Sep;21(11):3004-15. doi: 10.1096/fj.07-8203com. Epub 2007 May 3. FASEB J. 2007. PMID: 17478745 Free PMC article.
-
Increased expression of cyclin G1 and p21WAF1/CIP1 in neurons following transient forebrain ischemia: comparison with early DNA damage.J Neurosci Res. 1998 Aug 1;53(3):279-96. doi: 10.1002/(SICI)1097-4547(19980801)53:3<279::AID-JNR2>3.0.CO;2-7. J Neurosci Res. 1998. PMID: 9698156
-
Late-onset lipid peroxidation and neuronal cell death following transient forebrain ischemia in rat brain.Brain Res. 1997 Oct 24;772(1-2):37-44. doi: 10.1016/s0006-8993(97)00836-6. Brain Res. 1997. PMID: 9406953
-
Cerebral Edema After Cardiopulmonary Resuscitation: A Therapeutic Target Following Cardiac Arrest?Neurocrit Care. 2018 Jun;28(3):276-287. doi: 10.1007/s12028-017-0474-8. Neurocrit Care. 2018. PMID: 29080068 Review.
-
Pathophysiology and treatment of cerebral ischemia.J Med Invest. 1998 Aug;45(1-4):57-70. J Med Invest. 1998. PMID: 9864965 Review.
Cited by
-
Noninvasive delivery of gene targeting probes to live brains for transcription MRI.FASEB J. 2008 Apr;22(4):1193-203. doi: 10.1096/fj.07-9557com. Epub 2007 Nov 20. FASEB J. 2008. PMID: 18029447 Free PMC article.
-
Imaging C-Fos Gene Expression in Burns Using Lipid Coated Spion Nanoparticles.Adv J Mol Imaging. 2012 Oct;2(4):31-37. doi: 10.4236/ami.2012.24005. Adv J Mol Imaging. 2012. PMID: 24995147 Free PMC article.
-
MR contrast probes that trace gene transcripts for cerebral ischemia in live animals.FASEB J. 2007 Sep;21(11):3004-15. doi: 10.1096/fj.07-8203com. Epub 2007 May 3. FASEB J. 2007. PMID: 17478745 Free PMC article.
-
DNA-based MRI probes for specific detection of chronic exposure to amphetamine in living brains.J Neurosci. 2009 Aug 26;29(34):10663-70. doi: 10.1523/JNEUROSCI.2167-09.2009. J Neurosci. 2009. PMID: 19710318 Free PMC article.
-
Evaluation of neuronal protective effects of xanthine oxidoreductase inhibitors on severe whole-brain ischemia in mouse model and analysis of xanthine oxidoreductase activity in the mouse brain.Neurol Med Chir (Tokyo). 2015;55(1):77-85. doi: 10.2176/nmc.oa.2013-0307. Epub 2014 Dec 20. Neurol Med Chir (Tokyo). 2015. PMID: 25744353 Free PMC article.
References
-
- Roine RO, Raininko R, Erkinjuntti T, et al. Magnetic resonance imaging findings associated with cardiac arrest. Stroke. 1993;24:1005–14. - PubMed
-
- Fujioka M, Okuchi K, Sakaki T, et al. Specific changes in human brain following reperfusion after cardiac arrest. Stroke. 1994;25:2091–5. - PubMed
-
- Fujii M, Hara H, Meng W, et al. Strain-related differences in susceptibility to transient forebrain ischemia in SV-129 and C57black/6 mice. Stroke. 1997;28:1805–10. discussion 1811. - PubMed
-
- Kitagawa K, Matsumoto M, Yang G, et al. Cerebral ischemia after bilateral carotid artery occlusion and intraluminal suture occlusion in mice: evaluation of the patency of the posterior communicating artery. J Cereb Blood Flow Metab. 1998;18:570–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
