SNS-032 prevents tumor cell-induced angiogenesis by inhibiting vascular endothelial growth factor
- PMID: 17534442
- PMCID: PMC1877978
- DOI: 10.1593/neo.07136
SNS-032 prevents tumor cell-induced angiogenesis by inhibiting vascular endothelial growth factor
Abstract
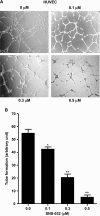
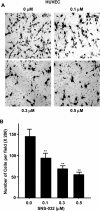
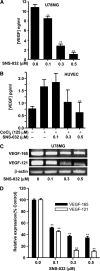
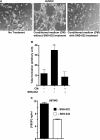
Cell proliferation, migration, and capillary network formation of endothelial cells are the fundamental steps for angiogenesis, which involves the formation of new blood vessels. The purpose of this study is to investigate the effect of a novel aminothiazole SNS-032 on these critical steps for in vitro angiogenesis using a coculture system consisting of human umbilical vein endothelial cells (HUVECs) and human glioblastoma cells (U87MG). SNS-032 is a potent selective inhibitor of cyclin-dependent kinases 2, 7, and 9, and inhibits both transcription and cell cycle. In this study, we examined the proliferation and viability of HUVECs and U87MG cells in the presence of SNS-032 and observed a dose-dependent inhibition of cellular proliferation in both cell lines. SNS-032 inhibited threedimensional capillary network formations of endothelial cells. In a coculture study, SNS-032 completely prevented U87MG cell-mediated capillary formation of HUVECs. This inhibitor also prevented the migration of HUVECs when cultured alone or cocultured with U87MG cells. In addition, SNS-032 significantly prevented the production of vascular endothelial growth factor (VEGF) in both cell lines, whereas SNS-032 was less effective in preventing capillary network formation and migration of endothelial cells when an active recombinant VEGF was added to the medium. In conclusion, SNS-032 prevents in vitro angiogenesis, and this action is attributable to blocking of VEGF.
Figures


Similar articles
-
SNS-032 prevents hypoxia-mediated glioblastoma cell invasion by inhibiting hypoxia inducible factor-1alpha expression.Int J Oncol. 2009 Apr;34(4):1051-60. doi: 10.3892/ijo_00000231. Int J Oncol. 2009. PMID: 19287962
-
Coptis japonica Makino extract suppresses angiogenesis through regulation of cell cycle-related proteins.Biosci Biotechnol Biochem. 2016 Jun;80(6):1095-106. doi: 10.1080/09168451.2016.1148574. Epub 2016 Feb 29. Biosci Biotechnol Biochem. 2016. PMID: 26924430
-
Antiangiogenic mechanisms of PJ-8, a novel inhibitor of vascular endothelial growth factor receptor signaling.Carcinogenesis. 2012 May;33(5):1022-30. doi: 10.1093/carcin/bgs127. Epub 2012 Mar 20. Carcinogenesis. 2012. PMID: 22436611
-
Senescence and its bypass in the vascular endothelium.Front Biosci. 2005 Jan 1;10:940-50. doi: 10.2741/1588. Print 2005 Jan 1. Front Biosci. 2005. PMID: 15569632 Review.
-
Endothelial cells on the move: dynamics in vascular morphogenesis and disease.Vasc Biol. 2020 Jul 2;2(1):H29-H43. doi: 10.1530/VB-20-0007. eCollection 2020. Vasc Biol. 2020. PMID: 32935077 Free PMC article. Review.
Cited by
-
CDK9 Blockade Exploits Context-dependent Transcriptional Changes to Improve Activity and Limit Toxicity of Mithramycin for Ewing Sarcoma.Mol Cancer Ther. 2020 May;19(5):1183-1196. doi: 10.1158/1535-7163.MCT-19-0775. Epub 2020 Mar 3. Mol Cancer Ther. 2020. PMID: 32127464 Free PMC article.
-
A SIX1 degradation inducer blocks excessive proliferation of prostate cancer.Int J Biol Sci. 2022 Mar 14;18(6):2439-2451. doi: 10.7150/ijbs.67873. eCollection 2022. Int J Biol Sci. 2022. PMID: 35414775 Free PMC article.
-
Cyclin-dependent kinase inhibitors in brain cancer: current state and future directions.Cancer Drug Resist. 2020 Mar 19;3(1):48-62. doi: 10.20517/cdr.2019.105. eCollection 2020. Cancer Drug Resist. 2020. PMID: 35582046 Free PMC article. Review.
-
Anticancer and Antiangiogenic Activities of Novel α-Mangostin Glycosides in Human Hepatocellular Carcinoma Cells via Downregulation of c-Met and HIF-1α.Int J Mol Sci. 2020 Jun 5;21(11):4043. doi: 10.3390/ijms21114043. Int J Mol Sci. 2020. PMID: 32516967 Free PMC article.
-
CDK Inhibition Primes for Anti-PD-L1 Treatment in Triple-Negative Breast Cancer Models.Cancers (Basel). 2022 Jul 11;14(14):3361. doi: 10.3390/cancers14143361. Cancers (Basel). 2022. PMID: 35884422 Free PMC article.
References
-
- Ferrara N, Gerber HP. The role of vascular endothelial growth factor in angiogenesis. Acta Haematol. 2001;106:148–156. - PubMed
-
- Frelin C, Ladoux A, D'Angelo G. Vascular endothelial growth factors and angiogenesis. Ann Endocrinol. 2000;61:70–74. - PubMed
-
- Brem S. Angiogenesis and cancer control: from concept to therapeutic trial. Cancer Control. 1999;6:436–458. - PubMed
-
- Carmeliet P. VEGF as a key mediator of angiogenesis in cancer. Oncology. 2005;69(Suppl 3):4–10. - PubMed
-
- Shih AH, Holland EC. Platelet-derived growth factor (PDGF) and glial tumorigenesis. Cancer Lett. 2006;232:139–147. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
