Ca2+-dependent inactivation of CaV1.2 channels prevents Gd3+ block: does Ca2+ block the pore of inactivated channels?
- PMID: 17535960
- PMCID: PMC2151623
- DOI: 10.1085/jgp.200709734
Ca2+-dependent inactivation of CaV1.2 channels prevents Gd3+ block: does Ca2+ block the pore of inactivated channels?
Abstract
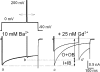
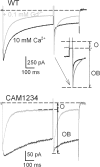
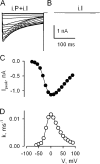
Lanthanide gadolinium (Gd(3+)) blocks Ca(V)1.2 channels at the selectivity filter. Here we investigated whether Gd(3+) block interferes with Ca(2+)-dependent inactivation, which requires Ca(2+) entry through the same site. Using brief pulses to 200 mV that relieve Gd(3+) block but not inactivation, we monitored how the proportions of open and open-blocked channels change during inactivation. We found that blocked channels inactivate much less. This is expected for Gd(3+) block of the Ca(2+) influx that enhances inactivation. However, we also found that the extent of Gd(3+) block did not change when inactivation was reduced by abolition of Ca(2+)/calmodulin interaction, showing that Gd(3+) does not block the inactivated channel. Thus, Gd(3+) block and inactivation are mutually exclusive, suggesting action at a common site. These observations suggest that inactivation causes a change at the selectivity filter that either hides the Gd(3+) site or reduces its affinity, or that Ca(2+) occupies the binding site at the selectivity filter in inactivated channels. The latter possibility is supported by previous findings that the EEQE mutation of the selectivity EEEE locus is void of Ca(2+)-dependent inactivation (Zong Z.Q., J.Y. Zhou, and T. Tanabe. 1994. Biochem. Biophys. Res. Commun. 201:1117-11123), and that Ca(2+)-inactivated channels conduct Na(+) when Ca(2+) is removed from the extracellular medium (Babich O., D. Isaev, and R. Shirokov. 2005. J. Physiol. 565:709-717). Based on these results, we propose that inactivation increases affinity of the selectivity filter for Ca(2+) so that Ca(2+) ion blocks the pore. A minimal model, in which the inactivation "gate" is an increase in affinity of the selectivity filter for permeating ions, successfully simulates the characteristic U-shaped voltage dependence of inactivation in Ca(2+).
Figures


Comment in
-
And yet it moves: conformational States of the Ca2+ channel pore.J Gen Physiol. 2007 Jun;129(6):457-9. doi: 10.1085/jgp.200709814. J Gen Physiol. 2007. PMID: 17535958 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

