Fast turnover of L1 adhesions in neuronal growth cones involving both surface diffusion and exo/endocytosis of L1 molecules
- PMID: 17538021
- PMCID: PMC1949362
- DOI: 10.1091/mbc.e06-12-1101
Fast turnover of L1 adhesions in neuronal growth cones involving both surface diffusion and exo/endocytosis of L1 molecules
Abstract
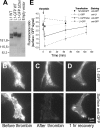
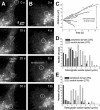
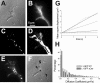
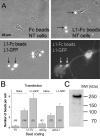
We investigated the interplay between surface trafficking and binding dynamics of the immunoglobulin cell adhesion molecule L1 at neuronal growth cones. Primary neurons were transfected with L1 constructs bearing thrombin-cleavable green fluorescent protein (GFP), allowing visualization of newly exocytosed L1 or labeling of membrane L1 molecules by Quantum dots. Intracellular L1-GFP vesicles showed preferential centrifugal motion, whereas surface L1-GFP diffused randomly, revealing two pathways to address L1 to adhesive sites. We triggered L1 adhesions using microspheres coated with L1-Fc protein or anti-L1 antibodies, manipulated by optical tweezers. Microspheres coupled to the actin retrograde flow at the growth cone periphery while recruiting L1-GFP molecules, of which 50% relied on exocytosis. Fluorescence recovery after photobleaching experiments revealed a rapid recycling of L1-GFP molecules at L1-Fc (but not anti-L1) bead contacts, attributed to a high lability of L1-L1 bonds at equilibrium. L1-GFP molecules truncated in the intracellular tail as well as neuronal cell adhesion molecules (NrCAMs) missing the clathrin adaptor binding sequence showed both little internalization and reduced turnover rates, indicating a role of endocytosis in the recycling of mature L1 contacts at the base of the growth cone. Thus, unlike for other molecules such as NrCAM or N-cadherin, diffusion/trapping and exo/endocytosis events cooperate to allow the fast renewal of L1 adhesions.
Figures
Similar articles
-
The role of endocytic l1 trafficking in polarized adhesion and migration of nerve growth cones.J Neurosci. 2001 Dec 1;21(23):9194-203. doi: 10.1523/JNEUROSCI.21-23-09194.2001. J Neurosci. 2001. PMID: 11717353 Free PMC article.
-
Regulation of N-cadherin dynamics at neuronal contacts by ligand binding and cytoskeletal coupling.Mol Biol Cell. 2006 Feb;17(2):862-75. doi: 10.1091/mbc.e05-04-0335. Epub 2005 Nov 30. Mol Biol Cell. 2006. PMID: 16319177 Free PMC article.
-
Semaphorin3A-induced receptor endocytosis during axon guidance responses is mediated by L1 CAM.Mol Cell Neurosci. 2004 May;26(1):89-100. doi: 10.1016/j.mcn.2004.01.010. Mol Cell Neurosci. 2004. PMID: 15121181
-
The mechanism of axon growth: what we have learned from the cell adhesion molecule L1.Mol Neurobiol. 2003 Dec;28(3):219-28. doi: 10.1385/MN:28:3:219. Mol Neurobiol. 2003. PMID: 14709786 Review.
-
Polarizing membrane dynamics and adhesion for growth cone navigation.Mol Cell Neurosci. 2011 Dec;48(4):332-8. doi: 10.1016/j.mcn.2011.03.007. Epub 2011 Apr 1. Mol Cell Neurosci. 2011. PMID: 21459144 Review.
Cited by
-
High N-glycan multiplicity is critical for neuronal adhesion and sensitizes the developing cerebellum to N-glycosylation defect.Elife. 2018 Oct 12;7:e38309. doi: 10.7554/eLife.38309. Elife. 2018. PMID: 30311906 Free PMC article.
-
Interplay between adhesion turnover and cytoskeleton dynamics in the control of growth cone migration.Cell Adh Migr. 2008 Oct-Dec;2(4):263-7. doi: 10.4161/cam.2.4.7274. Epub 2008 Oct 24. Cell Adh Migr. 2008. PMID: 19262147 Free PMC article.
-
Neurite outgrowth induced by NGF or L1CAM via activation of the TrkA receptor is sustained also by the exocytosis of enlargeosomes.Proc Natl Acad Sci U S A. 2014 Nov 25;111(47):16943-8. doi: 10.1073/pnas.1406097111. Epub 2014 Nov 10. Proc Natl Acad Sci U S A. 2014. PMID: 25385598 Free PMC article.
-
Role of the cytoplasmic domain of the L1 cell adhesion molecule in brain development.J Comp Neurol. 2010 Apr 1;518(7):1113-32. doi: 10.1002/cne.22267. J Comp Neurol. 2010. PMID: 20127821 Free PMC article.
-
Rabex-5 protein regulates the endocytic trafficking pathway of ubiquitinated neural cell adhesion molecule L1.J Biol Chem. 2012 Sep 21;287(39):32312-23. doi: 10.1074/jbc.M112.374322. Epub 2012 Jul 30. J Biol Chem. 2012. PMID: 22846990 Free PMC article.
References
-
- Brewer G. J. Thrombin causes cell spreading and redistribution of beta-amyloid immunoreactivity in cultured hippocampal neurons. J. Neurochem. 1996;67:119–130. - PubMed
-
- Brummendorf T., Lemmon V. Immunoglobulin superfamily receptors: cis-interactions, intracellular adapters and alternative splicing regulate adhesion. Curr. Opin. Cell Biol. 2001;13:611–618. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

