Interaction of the C-terminal region of the Ggamma protein with model membranes
- PMID: 17545235
- PMCID: PMC1965437
- DOI: 10.1529/biophysj.106.101196
Interaction of the C-terminal region of the Ggamma protein with model membranes
Abstract
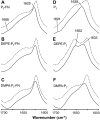
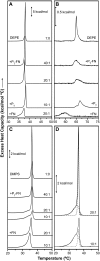
Heterotrimeric G-proteins interact with membranes. They accumulate around membrane receptors and propagate messages to effectors localized in different cellular compartments. G-protein-lipid interactions regulate G-protein cellular localization and activity. Although we recently found that the Gbetagamma dimer drives the interaction of G-proteins with nonlamellar-prone membranes, little is known about the molecular basis of this interaction. Here, we investigated the interaction of the C-terminus of the Ggamma(2) protein (P(gamma)-FN) with model membranes and those of its peptide (P(gamma)) and farnesyl (FN) moieties alone. X-ray diffraction and differential scanning calorimetry demonstrated that P(gamma)-FN, segregated into P(gamma)-FN-poor and -rich domains in phosphatidylethanolamine (PE) and phosphatidylserine (PS) membranes. In PE membranes, FN increased the nonlamellar phase propensity. Fourier transform infrared spectroscopy experiments showed that P(gamma) and P(gamma)-FN interact with the polar and interfacial regions of PE and PS bilayers. The binding of P(gamma)-FN to model membranes is due to the FN group and positively charged amino acids near this lipid. On the other hand, membrane lipids partially altered P(gamma)-FN structure, in turn increasing the fluidity of PS membranes. These data highlight the relevance of the interaction of the C-terminal region of the Ggamma protein with the cell membrane and its effect on membrane structure.
Figures





References
-
- Casey, P. J. 1995. Protein lipidation in cell signaling. Science. 268:221–225. - PubMed
-
- Sinensky, M. 2000. Functional aspects of polyisoprenoid protein substituents: roles in protein-protein interaction and trafficking. Biochim. Biophys. Acta. 1529:203–209. - PubMed
-
- Magee, T., and M. Hanley. 1988. Protein modification. Sticky fingers and CAAX boxes. Nature. 335:114–115. - PubMed
-
- Marshall, C. J. 1993. Protein prenylation: a mediator of protein-protein interactions. Science. 259:1865–1866. - PubMed
-
- Parish, C. A., and R. R. Rando. 1996. Isoprenylation/methylation of proteins enhances membrane association by a hydrophobic mechanism. Biochemistry. 35:8473–8477. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

