Synthesis of C5-dicarboxylic acids from C2-units involving crotonyl-CoA carboxylase/reductase: the ethylmalonyl-CoA pathway
- PMID: 17548827
- PMCID: PMC1965564
- DOI: 10.1073/pnas.0702791104
Synthesis of C5-dicarboxylic acids from C2-units involving crotonyl-CoA carboxylase/reductase: the ethylmalonyl-CoA pathway
Abstract
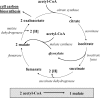
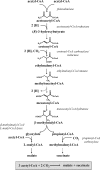
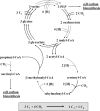
Fifty years ago, Kornberg and Krebs established the glyoxylate cycle as the pathway for the synthesis of cell constituents from C2-units. However, since then, many bacteria have been described that do not contain isocitrate lyase, the key enzyme of this pathway. Here, a pathway termed the ethylmalonyl-CoA pathway operating in such organisms is described. Isotopically labeled acetate and bicarbonate were transformed to ethylmalonyl-CoA by cell extracts of acetate-grown, isocitrate lyase-negative Rhodobacter sphaeroides as determined by NMR spectroscopy. Crotonyl-CoA carboxylase/reductase, catalyzing crotonyl-CoA + CO2 + NADPH --> ethylmalonyl-CoA- + NADP+ was identified as the key enzyme of the ethylmalonyl-CoA pathway. The reductive carboxylation of an enoyl-thioester is a unique biochemical reaction, unprecedented in biology. The enzyme from R. sphaeroides was heterologously produced in Escherichia coli and characterized. Crotonyl-CoA carboxylase/reductase (or its gene) can be used as a marker for the presence of the ethylmalonyl-CoA pathway, which functions not only in acetyl-CoA assimilation. In Streptomyces sp., it may also supply precursors (ethylmalonyl-CoA) for antibiotic biosynthesis. For methylotrophic bacteria such as Methylobacterium extorquens, extension of the serine cycle with reactions of the ethylmalonyl-CoA pathway leads to a simplified scheme for isocitrate lyase-independent C1 assimilation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Kornberg HL, Krebs HA. Nature. 1957;17:988–991. - PubMed
-
- Kornberg HL, Beevers H. Biochem Biophys Acta. 1957;26:531–537. - PubMed
-
- Patel TR, McFadden BA. Exp Parasitol. 1978;44:72–81. - PubMed
-
- Kornberg HL, Lascelles J. J Gen Microbiol. 1960;23:511–517. - PubMed
-
- Albers H, Gottschalk G. Arch Microbiol. 1976;111:45–49. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

