Heterologous prime/boost immunization of rhesus monkeys by using diverse poxvirus vectors
- PMID: 17553898
- PMCID: PMC1951337
- DOI: 10.1128/JVI.00744-07
Heterologous prime/boost immunization of rhesus monkeys by using diverse poxvirus vectors
Abstract
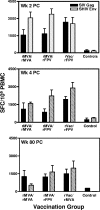
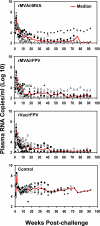
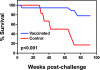
As the diversity of potential immunogens increases within certain classes of vectors, the possibility has arisen of employing heterologous prime/boost immunizations using diverse members of the same family of vectors. The present study was initiated to explore the use of divergent pox vectors in a prime/boost regimen to elicit high-frequency cellular immune responses to human immunodeficiency virus type 1 envelope and simian immunodeficiency virus gag in rhesus monkeys. We demonstrated that monkeys vaccinated with a recombinant modified vaccinia virus Ankara (rMVA) prime/recombinant fowlpox virus (rFPV) boost regimen and monkeys vaccinated with a recombinant vaccinia virus prime/rFPV boost regimen developed comparable cellular immune responses that were greater in magnitude than those elicited by a homologous prime/boost with rMVA. Nevertheless, comparable magnitude recall cellular immune responses were observed in monkeys vaccinated with heterologous and homologous recombinant poxvirus following challenge with the CXCR4-tropic SHIV-89.6P. Consistent with this finding, comparable levels of containment of viral replication and CD4(+) T-lymphocyte preservation were seen in these groups of recombinant poxvirus-vaccinated monkeys. This study supports further exploration of combining recombinant vectors of the same family in prime/boost immunization strategies to optimize vaccine-elicited cellular immune responses.
Figures



References
-
- Amara, R. R., F. Villinger, J. D. Altman, S. L. Lydy, S. P. O'Neil, S. I. Staprans, D. C. Montefiori, Y. Xu, J. G. Herndon, L. S. Wyatt, M. A. Candido, N. L. Kozyr, P. I. Earl, J. M. Smith, H. L. Ma, B. D. Grimm, M. L. Hulsey, J. Miller, H. M. McClure, J. M. McNicholl, B. Moss, and H. L. Robinson. 2001. Control of a mucosal challenge and prevention of AIDS by a multiprotein DNA/MVA vaccine. Science 292:69-74. - PubMed
-
- Barouch, D. H., S. Santra, J. E. Schmitz, M. J. Kuroda, T.-M. Fu, W. Wagner, M. Bilska, A. Craiu, X. X. Zheng, G. R. Krivulka, K. Beaudry, M. A. Lifton, C. E. Nickerson, W. L. Trigona, K. Punt, D. C. Freed, L. Guan, S. Dubey, D. Casimiro, A. Simon, M. E. Davies, M. Chastain, T. B. Strom, R. S. Gelman, D. C. Montefiori, M. G. Lewis, E. A. Emini, J. W. Shiver, and N. L. Letvin. 2000. Control of viremia and prevention of clinical AIDS in rhesus monkeys by cytokine-augmented DNA vaccination. Science 290:486-492. - PubMed
-
- Barouch, D. H., M. G. Pau, J. H. Custers, W. Koudstaal, S. Kostense, M. J. Havenga, D. M. Truitt, S. M. Sumida, M. G. Kishko, J. C. Arthur, B. Korioth-Schmitz, M. H. Newberg, D. A. Gorgone, M. A. Lifton, D. L. Panicali, G. J. Nabel, N. L. Letvin, and J. Goudsmit. 2004. Immunogenicity of recombinant adenovirus serotype 35 vaccine in the presence of pre-existing anti-Ad5 immunity. J. Immunol. 172:6290-6297. - PubMed
-
- Casimiro, D. R., L. Chen, T. M. Fu, R. K. Evans, M. J. Caulfield, M. E. Davies, A. Tang, M. Chen, L. Huang, V. Harris, D. C. Freed, K. A. Wilson, S. Dubey, D. M. Zhu, D. Nawrocki, H. Mach, R. Troutman, L. Isopi, D. Williams, W. Hurni, Z. Xu, J. G. Smith, S. Wang, X. Liu, L. Guan, R. Long, W. Trigona, G. J. Heidecker, H. C. Perry, N. Persaud, T. J. Toner, Q. Su, X. Liang, R. Youil, M. Chastain, A. J. Bett, D. B. Volkin, E. A. Emini, and J. W. Shiver. 2003. Comparative immunogenicity in rhesus monkeys of DNA plasmid, recombinant vaccinia virus, and replication-defective adenovirus vectors expressing a human immunodeficiency virus type 1 gag gene. J. Virol. 77:6305-6313. - PMC - PubMed
-
- Casimiro, D. R., F. Wang, W. A. Schleif, X. Liang, Z.-Q. Zhang, T. W. Tobery, M.-E. Davies, A. B. McDermott, D. H. O'Connor, A. Fridman, A. Bagchi, L. G. Tussey, A. J. Bett, A. C. Finnefrock, T. Fu, A. Tang, K. A. Wilson, M. Chen, H. C. Perry, G. J. Heidecker, D. C. Freed, A. Carella, K. S. Punt, K. J. Sykes, L. Huang, V. I. Ausensi, M. Bachinsky, U. Sadasivan-Nair, D. I. Watkins, E. A. Emini, and J. W. Shiver. 2005. Attenuation of simian immunodeficiency virus SIVmac239 infection by prophylactic immunization with DNA and recombinant adenoviral vaccine vectors expressing Gag. J. Virol. 79:15547-15555. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

